Development status
PAI-1 inhibitor: RS5614
Plasminogen activator inhibitor (PAI)-1 is a protein essential for blood clotting (thrombus). Therefore, the PAI-1 inhibitor we are developing has the effect of dissolving blood clots (fibrinolysis). However, when we conducted basic researches using our PAI-1 inhibitor, we confirmed its unexpected benefits such as 1) repairing cells caused by damage (cell regeneration), 2) preventing cellular aging (senescence), which makes cells unable to repair themselves over time, and 3) removing cancer cells. All of these effects lead to the extension of human healthy lifespan (longevity).
In developed countries, including Japan, the aging of the population is progressing, and the gap between average life expectancy and healthy life expectancy (the period of time when a person can live independently and in good physical and mental health, and is calculated by subtracting the period of being bedridden or in nursing care conditions such as dementia) from the average life expectancy has become a major issue. If we can treat the various diseases that occur with aging, such as cancer, cardiovascular disease, respiratory disease, and diabetes, we can extend our healthy life expectancy.These four diseases account for 70-80% of deaths worldwide, and are classified by the World Health Organization (WHO) as non-communicable diseases (NCDs), which are important diseases associated with aging and lifestyle habits. Our PAI-1 inhibitor has the potential to become a drug candidate for extending healthy life expectancy, including treatments for these four diseases.
R&D of PAI-1 inhibitor RS5614
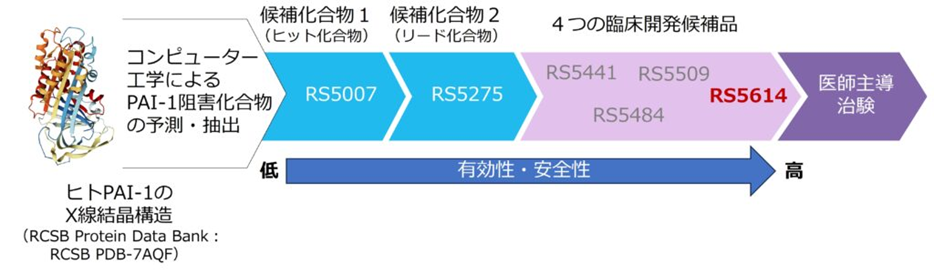
Based on the crystal structure of the human PAI-1 molecule and computer assisted drug design, we obtain 96 candidate compounds that theoretically inhibit human PAI-1 activity, from a library of approximately 2 million virtual compounds. We have synthesized more than 1,400 new derivatives over 10 years and evaluated their PAI-1 inhibitory activity and safety, resulting in the acquisition of RS5275, an orally administrable compound with excellent safety. Further synthesis development from RS5275 resulted in the acquisition of four clinical candidate compounds RS5441, RS5484, RS5509, and RS5614. Among them, we finally selected RS5614, which has the highest efficacy and safety, as a candidate compound for clinical development.
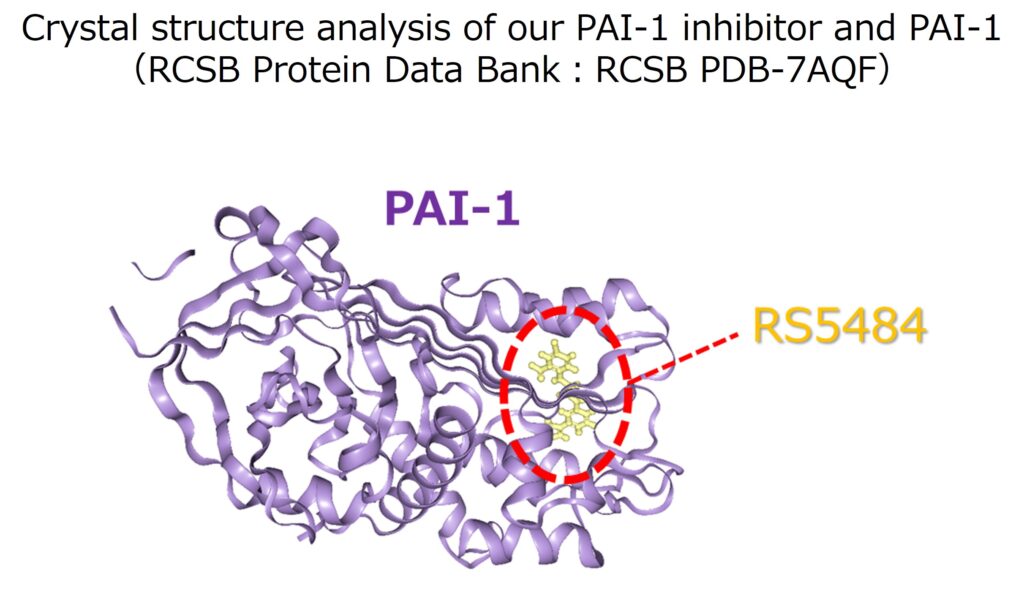
PAI-1 is a very unstable protein in the blood but is stabilizes after binding to vitronectin. As a result of X-ray crystal structure analysis, it is speculated that the developed PAI-1 inhibitor is inserted into the vitronectin binding site within the PAI-1 molecule, and by inhibiting the binding between PAI-1 and vitronectin, it inhibits the stabilization of the PAI-1 molecule and promotes its degradation.
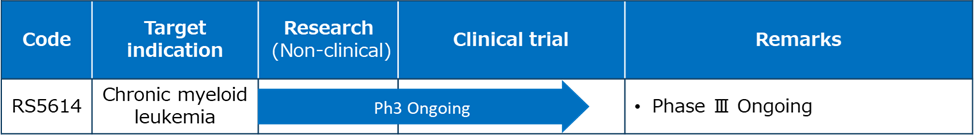
RS5614 has passed all non-clinical safety tests required for clinical trials and manufacturing and sales as a drug. In addition, RS5614 has been administered to nearly 400 subjects (healthy people, chronic myeloid leukemia, novel coronavirus lung injury, malignant melanoma, lung cancer, systemic sclerosis, etc.), and long-term administration for one year was also performed for chronic myeloid leukemia, but no problematic side effects due to RS5614 have been reported, and it is considered to be a highly safe drug. In the past, many pharmaceutical companies and bio start-ups, both in Japan and overseas, have been working on developing small molecule PAI-1 inhibitors, and some of the drugs have been reported to be effective in animal models of mice and rats. However, all of these were discontinued at an early stage of development, and our drug is the oral PAI-1 inhibitor currently in the human clinical stage.
RS5614 is currently under development for clinical applications in anti-aging/longevity, cancer, and lung diseases.
Anti-aging/longevity
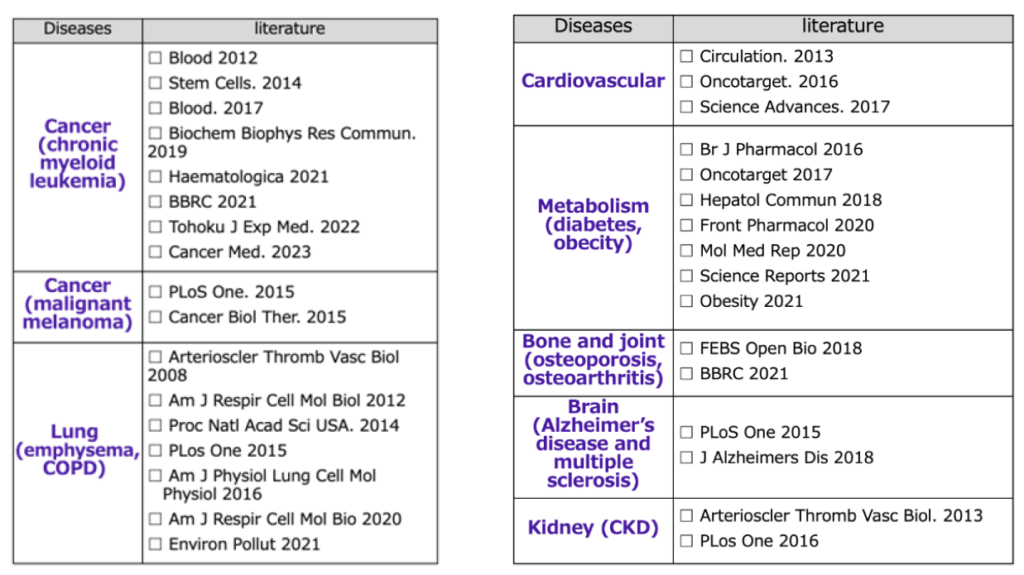
If we treat various age-related diseases in a multifaceted way, such as cancer, cardiovascular disease (arteriosclerosis), lung disease (emphysema, chronic obstructive pulmonary disease), metabolic disease (diabetes, obesity), chronic kidney disease, bone and muscle diseases (osteoporosis, sarcopenia), and cranial nerve diseases (cerebrovascular disease, Alzheimer’s disease, dementia), we can extend healthy lifespan.
Using our PAI-1 inhibitors, we have demonstrated the following series of scientific findings through research collaborations with Northwestern University and Tohoku University.
- Living cells cannot proliferate indefinitely due to a phenomenon called cellular senescence. This phenomenon involves shortening of gene telomere length and cellular senescence factors such as p53, p21, and p16ink4a. Senescent cells are known to have extremely high expression of plasminogen activator inhibitor (PAI)-1. The PAI-1 inhibitor developed by our company suppresses p53, p21, and p16ink4a, inhibiting cellular senescence in cardiomyocytes, fibroblasts, and vascular endothelial cells (Oncotarget, 2016).
- The PAI-1 inhibitor developed by our company reduces DNA damage in fibroblasts of Hutchinson-Gilford syndrome (designated intractable disease 333), a human premature aging disease, improves mitochondrial dysfunction, and corrects cellular abnormalities in Hutchinson-Gilford syndrome (Cell Death and Disease, 2022).
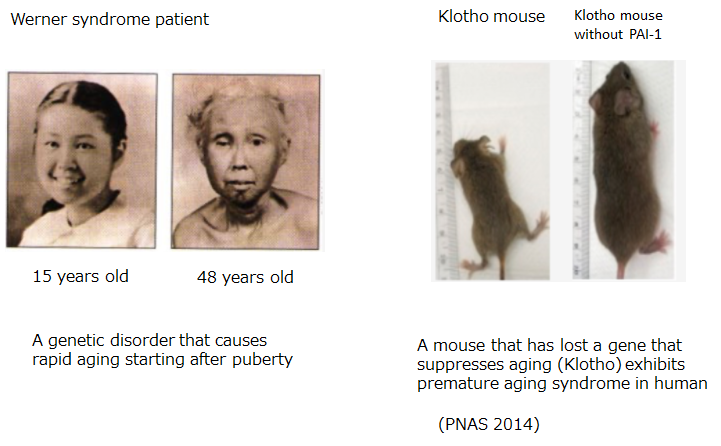
- It is known that PAI-1 expression is high not only in cells but also in aged tissues and individuals (mice and humans). In a non-clinical study using klotho mice, a well-known aging model, PAI-1 inhibitors improved the main symptoms of aging in klotho mice (Proc Natl Acad Sci USA. 2014).
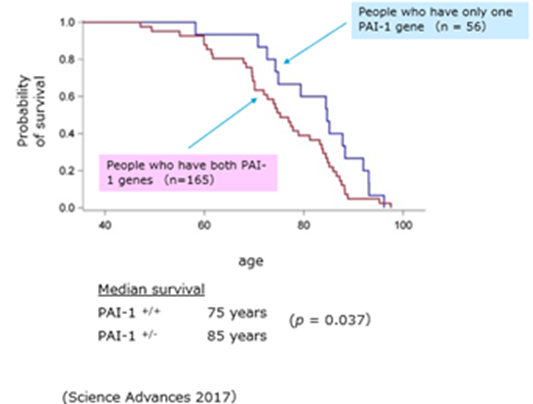
- A study was conducted on the blood of Amish people living in the United States, confirming that many people have a PAI-1 gene deficiency, and reporting that these PAI-1 gene deficiencies live about 10 years longer than those with the same gene (Science Advances, 2017). This epidemiological study in humans is consistent with the results of experiments in cells and mice. This fact was introduced in an article in the New York Times on November 21, 2017 (November 11, 2021).
- PAI-1 expression is extremely high in organs with age-related diseases such as cancer, cardiovascular disease (arteriosclerosis), lung disease (emphysema, chronic obstructive pulmonary disease), metabolic disease (diabetes, obesity), chronic kidney disease, bone and muscle disease (osteoporosis, sarcopenia), and cranial nerve disease (cerebrovascular disease, Alzheimer’s disease, dementia), and joint research with many universities both in Japan and overseas has revealed that administering our PAI-1 inhibitor can improve the pathology.
RS5614, which is currently under development, has been confirmed in non-clinical trials to have the potential to improve various age-related diseases, and is being tested in clinical trials for cancer and lung diseases. At this time, RS5614 is being developed as a medical drug (a drug used based on a doctor’s diagnosis or prescription, also known as a prescription drug), so clinical trials are being conducted for the regulatory approval for each individual disease.
Anti-aging and longevity benefits are difficult to verify in clinical trials, so they are not subject to prescription drugs. Meanwhile, anti-aging and longevity are important topics in the self-medication field and the OTC drug market, which are growing rapidly against the backdrop of a super-aging society. There is also an example of RS5441 being used as a hair loss treatment (anti-aging), and as our research into the anti-aging of our PAI-1 inhibitors progresses further, we would like to consider applications other than prescription drugs.
Pre-clinical studies on age-related disorders (published with our PAI-1 inhibitors)