Development status
Hematological Cancer: Therapeutic for Chronic Myeloid Leukemia (CML)
Chronic myeloid leukemia (CML), a type of hematological cancer, is caused by a genetic abnormality in the cells that give rise to blood cells (hematopoietic stem cells) that results in the unrestricted growth of cancerous leukemia cells (CML cells).
Current Treatments and Challenges
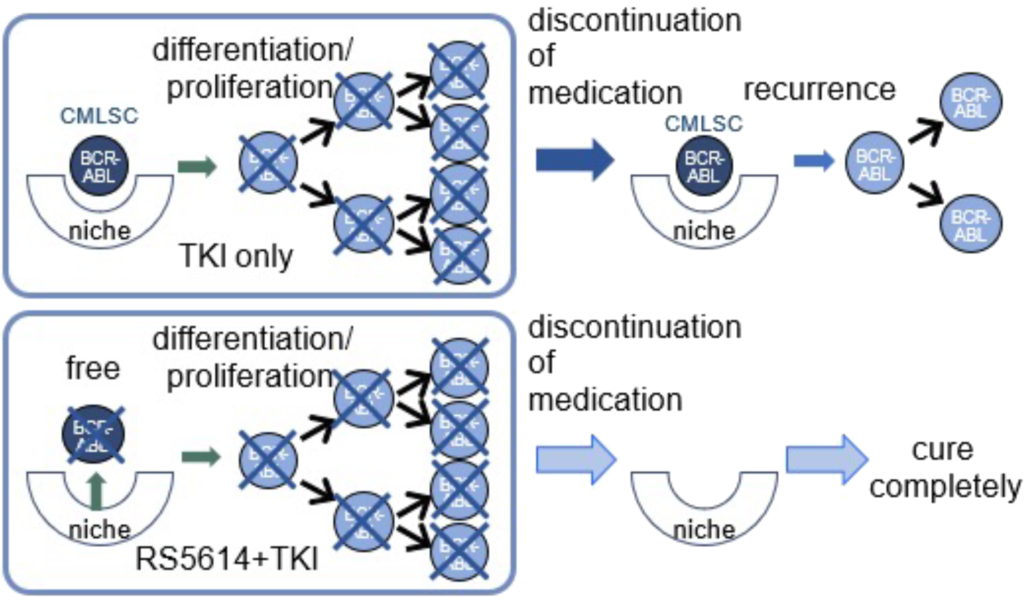
The mainstay of treatment for CML is the anticancer agent tyrosine kinase inhibitor TKI (e.g., imatinib) The development of TKI has greatly improved the survival of patients with CML. However, TKI act on CML cells but not on the cells that give rise to CML cells (CML stem cells), which hide in a site in the bone marrow called the “bone marrow niche,” so that when TKI is withdrawn, the CML stem cells turn into CML cells and relapse. Therefore, long-term, expensive TKI treatment must be continued to cure CML, and its side effects are also a problem. Therefore, it is important to achieve complete cure without the need to take TKI as early as possible.
It has recently been shown that CML patients who have been in a state of almost no detectable CML gene abnormalities in their blood (called “deep remission, DMR”) for a certain period of time can achieve relapse-free status even after TKI discontinuation. However, the percentage of patients who achieve relapse-free status within a minimum treatment period of 3 years is only 5-10%. Furthermore, the condition to achieve relapse-free status requires maintenance of DMR for at least two years after achieving DMR.
Characteristics of Our Solution
Our drug acts on CML stem cells and releases them from the bone marrow niche. The released CML stem cells are transformed into CML cells that are susceptible to TKI, and it has been shown that CML stem cells in the bone marrow niche are eliminated, resulting in the complete cure of CML. In fact, when RS5614 was combined with a TKI (imatinib) in a mouse model of CML, the number of CML stem cells remaining in the bone marrow was markedly reduced and survival was greatly improved compared to imatinib alone.
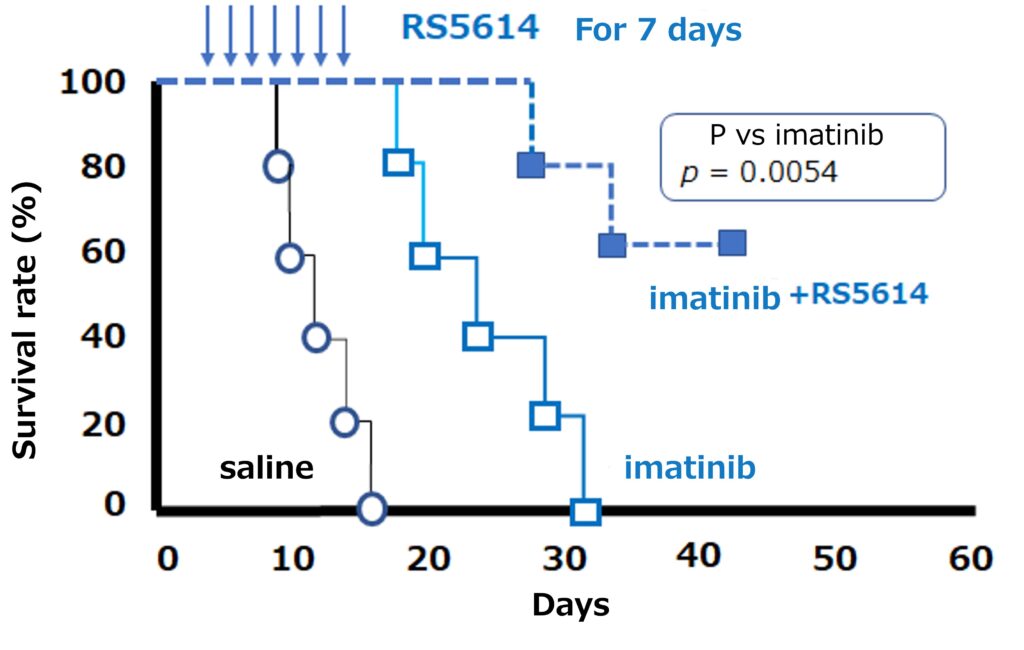
RS5614 is expected to be a safe drug with a new mechanism of action that, when combined with TKI, leads to early and complete cure of many CML patients, rendering drug therapy unnecessary.
Results to date
Early phase II study (Ph2a)
A Phase II investigator-initiated clinical trial was conducted at Tohoku University, Tokai University, and Akita University for CML patients who had been treated with TKI for at least 2 years. Twenty-one (21) CML patients were treated with TKI and RS5614 (120 mg once daily) for 4 weeks, and the status that almost no abnormal CML genes were detected in the blood (DMR) was evaluated after 12 weeks. As a result, while only 2.0% of the patients could achieve DMR after 12 weeks in the treatment with TKI alone (published data, called “historical control”), 4 out of 21 patients treated with RS5614 in combination with TKI achieved DMR, and the cumulative DMR achievement rate at 12 weeks was 20.0%. In terms of safety, there were no adverse drug reactions caused by RS5614 in all 21 patients included in the analysis.
Late Phase II Study (Ph2b)
An investigator-initiated clinical trial was conducted at Tohoku University, Tokai University, and Akita University in 33 patients with CML, in which TKI and RS5614 (150 mg once daily, the dose can be increased to 180 mg) were administered concomitantly for 48 weeks, and DMR achievement rate was evaluated as an endpoint. The results showed that the cumulative DMR achievement rate was 33.3% (11 of 33 patients), which was significantly higher than the historical control rate of 8.0% obtained with 48 weeks of TKI-only treatment, confirming the efficacy of RS5614. In terms of safety, there was no serious side effects caused by RS5614.
The results of this study were published in the scientific journal “Cancer Medicine”.
[Results of Phase II studies of RS5614 for CML]
| Study | Subject number | Duration of RS5614 administration | Cumulative DMR achievement rate | Historical control |
| Ph2a | 21 | 4 weeks | 20.0% | 2% |
| Ph2b | 33 | 48 weeks | 33.3% | 8% |
Progress to date
Based on the results of the late-stage Phase II trial, with support from the Japan Agency for Medical Research and Development (AMED) “Innovative Cancer Medical Practical Research Project,” a double-blinded, placebo-controlled Phase III investigator-initiated clinical trial that compares a combination of TKI and RS5614 with a combination of TKI and placebo (placebo) has been conducted since March 2022, in collaboration with 12 medical institutions, including Tohoku University, Tokai University, and Akita University, in patients with chronic-phase CML with a TKI treatment period of 3 years or more. In order to eliminate drug treatment in CML patients, DMR must be maintained for 2 years or more, and we will verify whether TKI plus RS5614 significantly increases DMR compared to TKI plus placebo. As of the end of December 2023, 57 cases, exceeding the number of cases required for analysis, were registered. In addition, as a result of the final year evaluation of AMED’s “Innovative Cancer Medical Practical Research Project” conducted in December 2024, a further two-year extension of the grant period was approved, on the grounds that the target number of cases for the Phase III trial has been registered and the trial is on track to be completed within the two-year extension period.