Development status
Respiratory Disease: Therapeutic for Interstitial Lung Disease Associated with Systemic Scleroderma
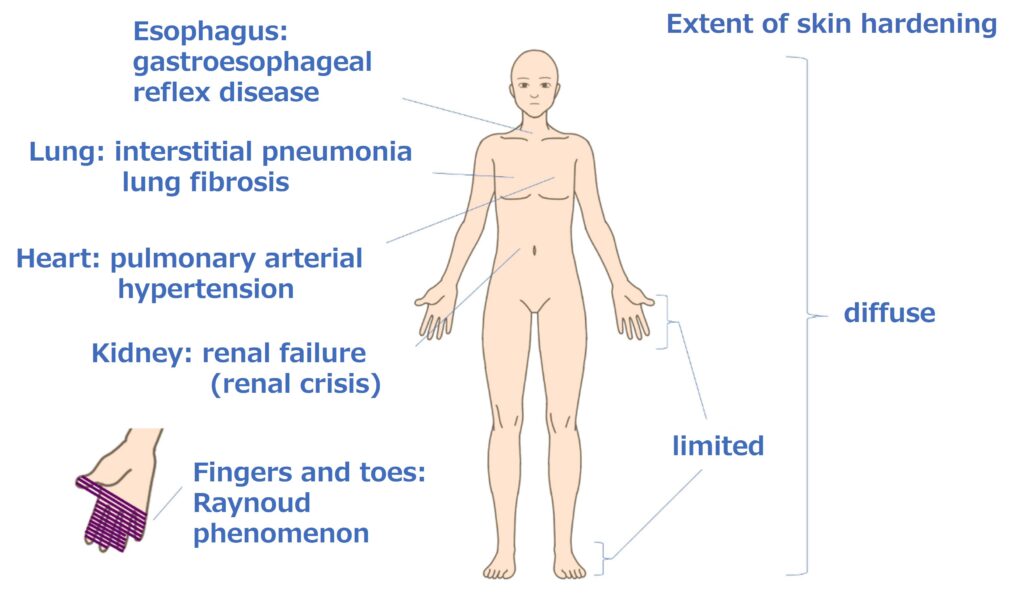
Systemic scleroderma is a systemic autoimmune disease characterized by vasculopathy and fibrosis that hardens the skin and many organs, and the intractable disease designated by the government based on the ” Act on Medical Care for Patients with Intractable Diseases” (Designated intractable diseases No 51).
More than 20,000 patients have been confirmed in Japan, but the cause is unknown and there is no curative treatment.
Systemic sclerosis has three main pathologies: “immune abnormalities,” “vascular disorders,” and “fibrosis.” Clinical symptoms due to organ fibrosis include Raynaud’s syndrome1), skin sclerosis, interstitial lung disease, scleroderma renal crisis2), cardiac lesions, and pulmonary arterial hypertension3), which cause various multi-organ disorders. Compared to other autoimmune diseases, the effects of steroids and immunosuppressants are limited. Interstitial lung disease in particular accounts for 35 % of deaths, and even in cases where interstitial lung disease is not the direct cause of death, severe respiratory function decline leads to a significant decline in quality of life (QOL) and activities of daily living (ADL). Steroids and immunosuppressants are considered the first-choice drugs for interstitial lung disease, but their therapeutic effects are limited. In addition, nintedanib, an anti-fibrotic drug4), has been approved, but its effect is limited to inhibiting the progression of interstitial lung disease and no improvement has been observed. Therefore, there is a strong need for the development of a new therapeutic drug that can improve interstitial lung disease associated with systemic sclerosis.
In pre-clinical trials, RS5614 has been confirmed to suppress inflammation, vascular disorders, thrombosis, and fibrosis (including pulmonary fibrosis). When RS5614 was orally administered daily to an animal model of interstitial lung disease associated with systemic sclerosis, RS5614 dose-dependently reduced pulmonary fibrosis, and its effect was superior to that of the existing therapeutic drug nintedanib, making it a promising new therapeutic drug. In addition, the efficacy of RS5614 as a treatment for lung injury was suggested in a Phase II investigator-initiated clinical trial of novel coronavirus lung injury conducted in 2020, and its safety was confirmed.
From October 2023, a placebo-controlled double-blind study5) to confirm the efficacy and safety of RS5614 for interstitial lung disease associated with systemic sclerosis was started at medical institutions of 10 domestic universities, such as Tohoku University, University of Tokyo, Kanazawa University, Fukui University, Osaka University, Wakayama Medical University, Gunma University, Yokohama City University, Sapporo Medical University, and Fujita Health University, targeting 50 patients with interstitial lung disease associated with systemic sclerosis. The target number of cases, 50 cases, was registered in December 2024 and is currently underway. This study has been selected by the AMED as “Research Project for Practical Application of Intractable Diseases (representative institution: Tohoku University, our company is a contributing institution)”.
1)Raynaud’s syndrome
This symptom is when the fingers turn pale to purple when touching something cold. It is often seen in winter and is the most common initial symptom.
2)Scleroderma renal crisis
This is a condition in which damage to the blood vessels of the kidneys occurs, resulting in high blood pressure. Along with the sudden rise in blood pressure, headaches and nausea also occur.
3)Pulmonary arterial hypertension
To live, humans need to breathe and take in oxygen from the air into the lungs, but breathing alone with the lungs is not enough to take in oxygen into the body. The oxygen taken into the lungs needs to be returned to the heart and then sent to the whole body. The blood vessels that send blood from the heart to the lungs are called pulmonary arteries. Pulmonary arterial hypertension is when the blood pressure in these pulmonary arteries rises abnormally. The reason the pressure in the pulmonary arteries rises is because the small blood vessels in the lungs become abnormally narrow and hard, which causes poor blood flow. In order to send the necessary oxygen to the body, it is necessary to keep the amount of blood leaving the heart at a certain level. The heart makes an effort to force blood through the narrow blood vessels, which causes the blood pressure in the pulmonary arteries to rise. Pulmonary arterial hypertension is designated as an intractable disease.
4)Antifibrotic drugs
As the name suggests, these drugs suppress tissue fibrosis. They may be prescribed to patients who are diagnosed with fibrosis or who are expected to develop fibrosis in the future. There are two types of antifibrotic drugs: pirfenidone and nintedanib.
5)Placebo-controlled double-blind
This is a clinical trial method in which patients are randomly divided into a group that receives the investigational drug (RS5614 in this case) and a group that receives a control drug (an ineffective placebo in this case), and both groups are administered the drug at the same time, with neither the doctor nor the patient knowing which group they will be administered. This is a test method to reduce the opportunity for doctors to administer the investigational drug to patients who are expected to benefit from it, and to avoid the possibility of preconceived notions that the drug should be effective being reflected in the evaluation, or the possibility that the patient’s reaction to the treatment or evaluation will be affected if they know about it.