Development status
Development of Software as a Medical Device Utilizing Artificial Intelligence (AI)
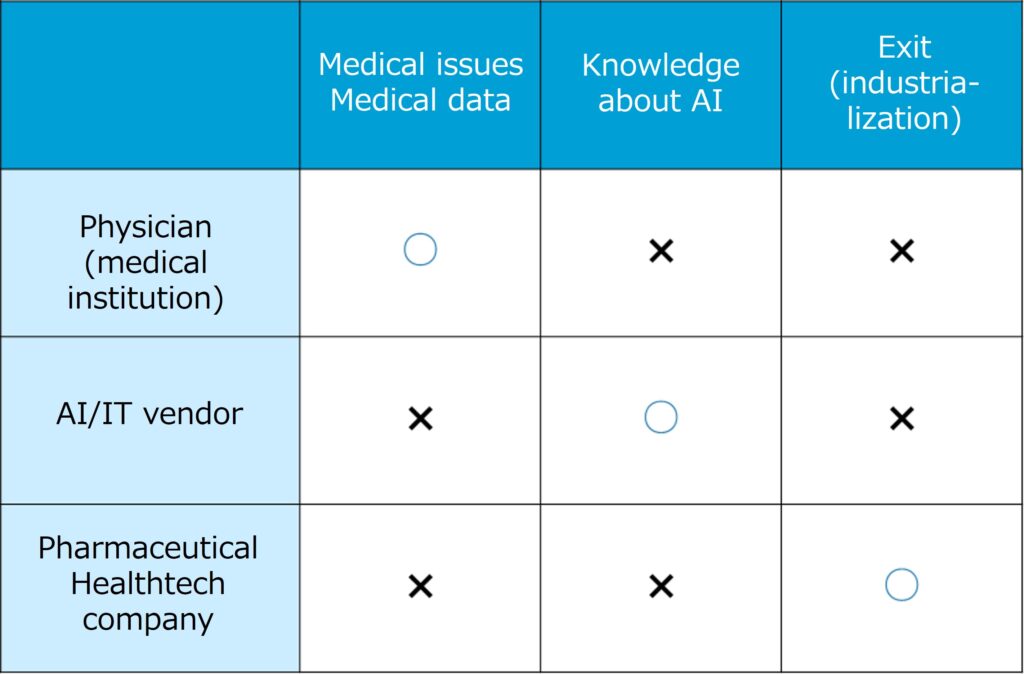
The application of artificial intelligence (AI) to the medical field is a theme with great potential, but the stakeholders who play an important role in research and development of medical AI face individual challenges. Although physicians (medical institutions) are well versed in medical issues and problems (needs) and possess a wealth of medical data and ideas, they lack AI technologies and networks with IT vendors, making it difficult for them to launch specific R&D projects. On the other hand, IT vendors with AI technologies are interested in applying them to the medical field, which is expected to grow, but practical application is not easy because they have little network with physicians (medical institutions), making it difficult to access medical needs and medical data, and they lack experience in pharmaceutical administration and regulations, including the Pharmaceutical and Medical Device Act.
Furthermore, pharmaceutical and healthtech companies, at the exit of the R&D, that wish to commercialize medical applications of AI often find it difficult to handle everything from research to business development on their own, both in terms of time and resources. Therefore, it is important to have a framework in which physicians (medical institutions) who has medical issues, IT vendors with AI technology, and exit pharmaceutical/healthtech companies can collaborate from the beginning to promote the development of medical AI.
It is essential to plan efficient research and development from the early stages of development with an eye toward the regulatory approval and the clinical implementation, since the product life cycle of AI-based medical solutions is not as long as that of pharmaceuticals. For this reason, open innovation among different fields of science is important, and data scientists, AI researchers, and pharmaceutical affairs experts must work together in addition to physicians. In the process of conducting many investigator-initiated clinical trials, Renascience has established networks with numerous medical institutions and multiple medical departments, and has easy access to medical issues and medical data (medical support). Renascience has concluded collaborative business agreements with multiple IT companies through open innovation (technological support). Renascience can also support the pharmaceutical administration and regulations through experiences of conducting investigator-initiated clinical trials for pharmaceuticals.
Therefore, we have been working as a hub that links physicians at medical institutions, IT vendors with AI technology, and pharmaceutical/healthtech companies for industrialization. We work to build an ecosystem that connects AI research to business in the medical field. Since we can conduct clinical trials in accordance with the Pharmaceutical Affairs Law, it will be possible for us to develop full-scale medical solutions (diagnosis and treatment) that can be used in actual clinical practice.
Computer programs (software) for the purpose of diagnosis, treatment, etc. are newly positioned as medical devices under a 2013 amendment to the law. These programs are installed in computers and mobile terminals, etc., and the recording media on which such programs are installed are “software as a medical device”. Renascience develops various software as a medical device utilizing AI through collaborations with an external network.
The essential elements for the R&D of medical AI are 1) medical issues, 2) medical data (quality and quantity), and 3) AI algorithms (engines). Many companies that possess the technology have faced significant challenges, in which even excellent technologies are difficult to put to practical use because they do not meet the needs of the medical field. In recent years, the “Biodesign” method has attracted attention, in which solutions are developed starting from the needs of the medical field and optimized by envisioning the final product in the medical field. The same applies to software as a medical device (SaMD) that utilizes AI as their core technology; AI technology is of course important, but the objective of medical data is not a machine but a patient with significant differences from individual to individual. Therefore, it is important to customize the AI based on each medical issue, each set of medical data, and the advice by physicians at the medical settings.
The essential material for AI research is medical data; although AI requires a sufficient amount of data to discover rules from a large amount of data (inductive inference), the quality of data is also important in addition to the quantity of data. In addition, the optimal AI algorithm should be selected based on the medical problem to be solved and the type of medical data to be utilized. Rather than looking for a medical field where a specific AI algorithm can be utilized, it is necessary to “select (or in some cases, develop independently) the optimal AI algorithm” to solve a specific medical issue. In addition, data scientists can analyze the existing medical data with AI after the AI algorithm has been determined, but only physicians can interpret and judge whether the results are correct. Therefore, without physician involvement, good quality medical data cannot be utilized to train AI to solve problems. Collaboration among physicians, data scientists, and AI researchers is important, and active involvement of the medical field (physicians), where medical issues, medical data, experience, and knowledge exist, is the key to success in solving medical issues through AI.
To promote research and development on medical applications of AI, we signed a memorandum of understanding (MOU) with NEC Solution Innovator, Ltd. (NES) in November 2022 and a comprehensive collaboration agreement (MOU) with NEC Corporation in June 2023.
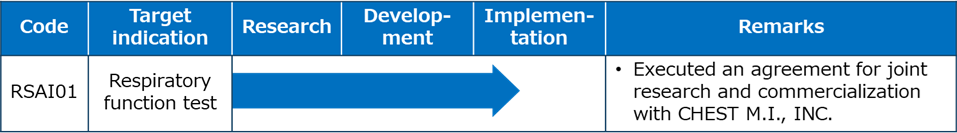
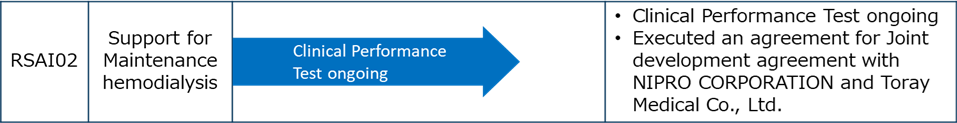
The development of software as a medical device utilizing AI requires a core AI (software) and a system to run it in the medical field. The most appropriate AI must be selected based on the medical issue and the type of medical data. We have licensed several basic AIs (engines) from NEC and customized them for medical use at medical settings. We have been also developing a system with NES to utilize the developed medical AI in the medical field.
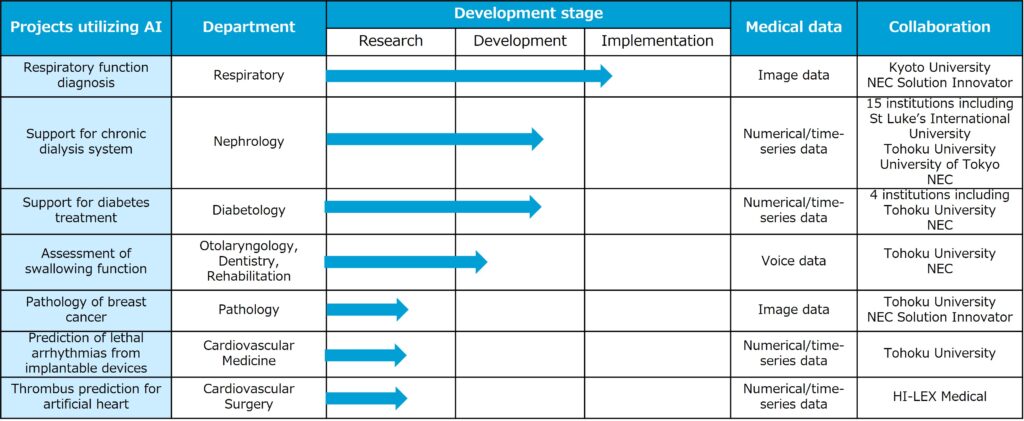
As a project in the exploratory stage, we develop an AI to detect lesions from pathological images of breast cancer in collaboration with Tohoku University, as part of the efforts on our priority areas, women’s health. In the verification studies using the pathological images, the detection model was classified into three classes (benign, non-invasive cancer, and invasive cancer) or two classes (benign and malignant), and achieved diagnostic accuracy of 88.3% and 90.5%, respectively. In the future, we plan to work on AI diagnosis using “intraoperative rapid pathology specimens” in the breast cancer.
In addition, as an initiative related to aging-related diseases, we develop, in collaboration with Tohoku University, an AI that predicts the onset of heart failure and fatal arrhythmias in advance by utilizing remote monitoring information of patients with cardiac implantable electrical devices.
Furthermore, in September 2022, we entered into a collaboration agreement with HI-LEX Corporation and HI-LEX Medical Corporation, and are working with HI-LEX Medical Corporation and Tohoku University to develop AI to predict the occurrence of blood clots in auxiliary artificial hearts.