Development status
Solid cancer: Therapeutic for Malignant melanoma
Malignant melanoma is a tumor of pigment-producing cells called melanocytes in the epidermis and is considered extremely malignant with a high metastasis rate among skin cancers. According to a survey conducted by the Japanese Association of Clinical Cancer Centers (2010-2014), the 5-year survival rate is 54.7% for stage 3 (stage with metastasis in lymph nodes or surrounding skin or subcutaneous sites) and 8.3% for stage 4 (stage with metastasis in other organs), making the disease extremely difficult to treat.
Current Treatments and Challenges
Though malignant melanoma has been a highly malignant and difficult-to-treat disease, the approval of the immune checkpoint inhibitor anti-PD-1 antibody in 2014 and the subsequent development of new drugs have led to a breakthrough in pharmacotherapy.
The immune checkpoint inhibitors are novel therapeutics developed as cancer immunotherapy, including anti-PD-1 antibody as an antibody drug for PD-1(nivolumab, trade name: Opdivo) against “the brake” on the immune system (immune checkpoint molecule) PD-1 discovered by Dr Tasuku Honjo and his team at Kyoto University (Dr. Tasuku Honjo was awarded the Nobel Prize in physiology or medicine in 2018). Immune checkpoint inhibitors release “the brake” on the immune system (immune checkpoint molecule) so that the body’s natural immunity can attack tumors.
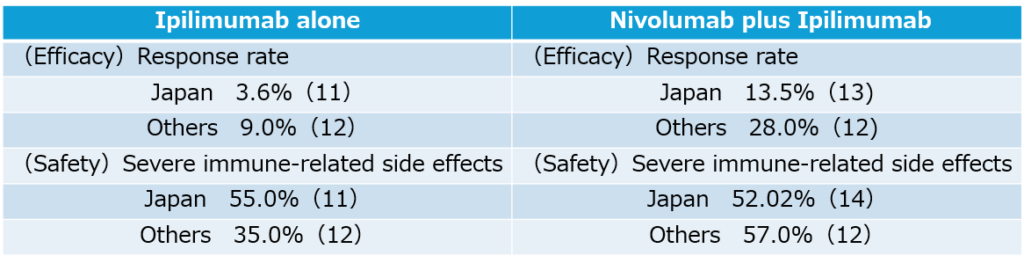
Since a major type of malignant melanoma in Japan (called “Acral Lentiginous Melanoma”) do not respond well to nivolumab unlike in Europe and the United States, the effectiveness of nivolmab is not sufficient, and the second-line treatment for the patients refractory to nivolmab is eagerly anticipated. Currently, another immune checkpoint inhibitor, the anti-CTLA-4 antibody ipilimumab, is approved as a concomitant use with nivolumab, and the nivolumab plus ipilimumab combination therapy has shown a higher response rate (13.5%) compared to the nivolumab alone. However, this combination therapy has also become a social problem, as more than half of patients experience serious side effects, and as the frequency of severe immune-related side effects that result in discontinuation of treatment is four times higher than with single treatment with nivolumab, requiring several months of hospitalization and cessation of cancer treatment. In addition, the use of two antibodies results in higher medical costs, so there is a need for a new second-line treatment that is safe, inexpensive, and convenient.
<Differences in drug responses between Japan and other countries>
Characteristics of Our Solution
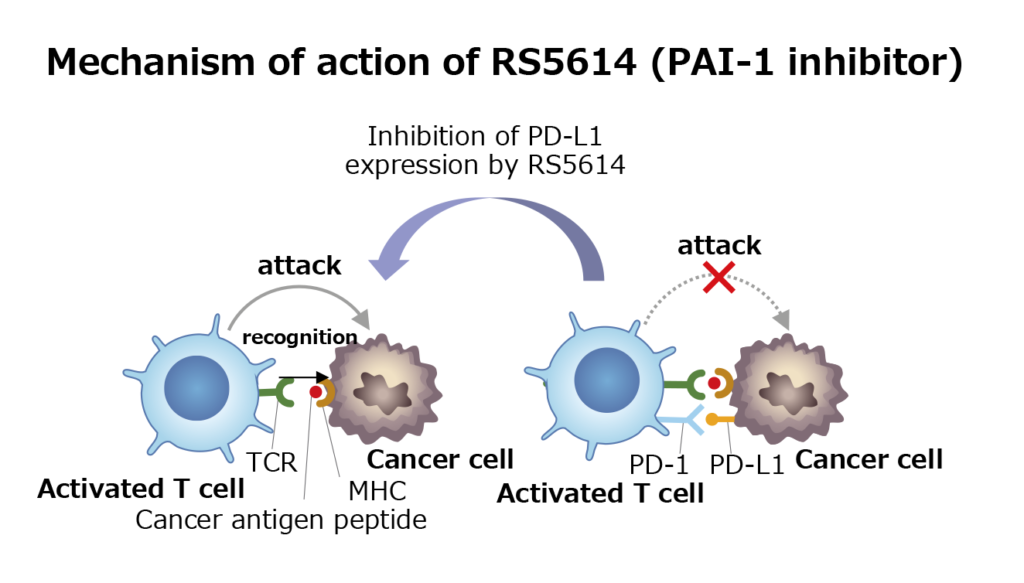
We have discovered that PAI-1 suppresses immunity to cancer via immune checkpoint molecules. In addition, studies in animal models have shown that administration of RS5614 inhibits the growth of tumors such as malignant melanoma and colorectal cancer. Furthermore, while the immune checkpoint inhibitor anti-PD-1 antibody alone inhibited tumor growth, the combination of RS5614 with the anti-PD-1 antibody enhanced cancer immunity strongly. As a result, the combination of these drugs strongly inhibited the growth of various cancers, and some types of tumors regressed. The PAI-1 inhibitor improve the immune environment in tumors to facilitate the action of the anti-PD-1 antibody.
While side effects have been a problem with the existing nivolumab + ipilimumab combination therapy, RS5614 will be a safe and convenient medication that can be taken at home. In addition, unlike antibodies, RS5614 is chemically synthesized, so its price is expected to be lower than that of antibodies.
Phase II clinical trial
A Phase II clinical trial aimed at confirming the tolerability and safety of the combination of TM5614 and nivolumab in patients with malignant melanoma (unresectable and unresponsive to anti-PD-1 antibodies) was conducted as a multicenter, open-label, investigator-initiated trial at seven university hospitals nationwide, i.e, Tohoku University, University of Tsukuba, Tokyo Metropolitan Cancer and Infectious Diseases Center Komagome Hospital, Kinki University, Nagoya City University, and Kumamoto University, which belong to an NPO, Japan Skin Cancer Network (JSCaN), which are collaborating to improve treatment outcomes for malignant melanoma between September 2021 and March 2023.
As a result of this clinical trial, 29 patients with malignant melanoma were treated with the PAI-1 inhibitor RS5614 developed by our company for 8 weeks, and 7 patients showed a response in the primary endpoint (response rate 24.1%).
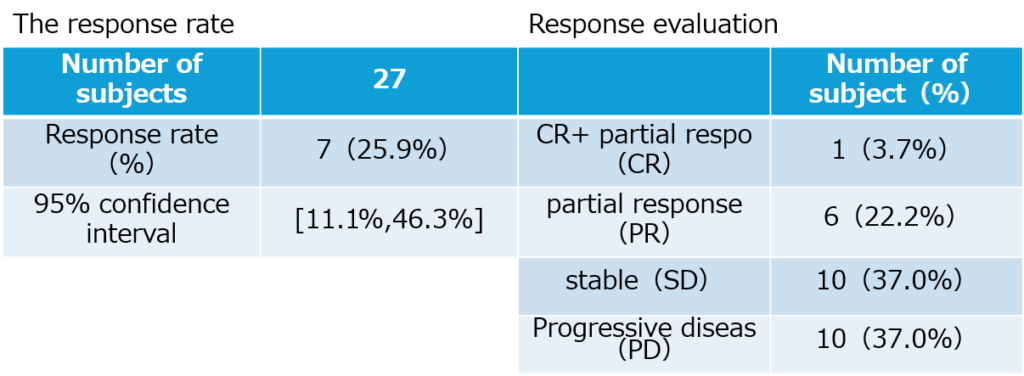
This response rate was equal to or better than the efficacy of the currently approved combination of nivolumab and ipilimumab (response rate of nivolumab and ipilimumab combination in nivolumab ineffective cases was 21% overseas and 13.5% in Japan). In addition, the disease control rate with the combination of nivolumab and RS5614 reached 62%. The combination of nivolumab and ipilimumab has been problematic due to the frequent occurrence of severe immune-related side effects, but the combination of nivolumab and RS5614 did not show any particularly problematic severe side effects. A clinical trial report was prepared in February 2024, and the results of this clinical trial were published in the scientific journal “British Journal of Dermatology” in June 2024.
Phase III clinical trial
Based on the results of the Phase II clinical trial, the Ministry of Health, Labor and Welfare designated the drug as an orphan disease drug for malignant melanoma in August 2024. This designation is expected to result in preferential guidance and advice from the PMDA, a marketability premium in drug price calculations, and an extended reexamination period after approval, which will extend the monopoly period of this therapeutic drug business. In addition, there is a possibility that preferential treatment such as subsidies will be available through the National Institutes of Biomedical Innovation, Health and Nutrition.
On February 18, 2025, a Phase III trial with RS5614 for malignant melanoma have started. This clinical trial is a randomized, placebo-controlled, double-blind, investigator-initiated phase III syudy (scheduled registration period 1.5 years, planned observation period 3 years) to verify the efficacy and safety of the combination of nivolumab and RS5614 in 124 patients with unresectable malignant melanoma, and will be conducted at 18 facilities in Japan, including Tohoku University Hospital (Tohoku University Hospital, Sapporo Medical University Hospital, Hirosaki University Hospital, Jichi Medical University Saitama Medical Center, National Cancer Center Hospital East, National Cancer Center Hospital, Cancer Institute Ariake Hospital, Niigata Cancer Center Niigata Hospital, Shizuoka Cancer Center, Nagoya City University Hospital, University of Tsukuba Hospital, Gifu University Hospital, Shimane University Hospital, Ehime University Hospital, Kyushu University Hospital, Kyushu Cancer Center, Kumamoto University Hospital, and Kagoshima Medical Center). The primary endpoint is overall survival (O complete response S).