Business Model
Research and Development strategy
Business Portfolio
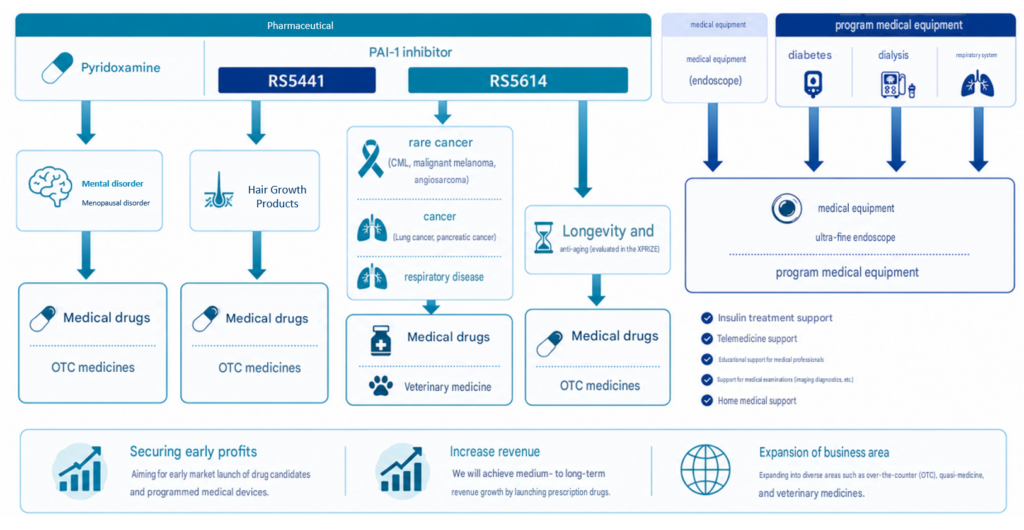
Our company has two main business portfolios: pharmaceuticals and medical devices/programmed medical devices. This is because we aim to reduce risk and achieve early profitability and expand future earnings. The pharmaceutical business is an area with high business risk due to large research and development (R&D) costs and periods, but we can expect high earnings after launch. On the other hand, the medical device and software as a medical device (SaMD) businesses are smaller than pharmaceuticals, but the R&D costs and periods and business risks are small, so they will lead to earnings for our company relatively quickly.
Cancer, anti-aging, and longevity
In the pharmaceutical field, we are primarily developing the PAI-1 inhibitor RS5614 (cancer and anti-aging/longevity) and RS5441 (topical treatment for male pattern baldness and age-related alopecia). The PAI-1 inhibitor RS5614 activates the immune system and promotes the removal of cancer cells and senescent cells, as well as possessing a variety of other effects, including antithrombotic, anti-inflammatory, and anti-fibrotic properties. Clinical trials for several cancers are currently underway in Japan (Phase III for chronic myeloid leukemia and malignant melanoma, Phase II for angiosarcoma and lung cancer). We aim to first obtain regulatory approval in Japan for rare cancers (malignant melanoma, angiosarcoma, and chronic myeloid leukemia) in order to launch the drug and applying it clinically. In parallel, we plan to conduct Phase II trials to expand the indications to lung cancer, pancreatic cancer, and other major cancers, thereby securing a large future market.
We are also developing the PAI-1 inhibitor RS5614 in the field of pulmonary diseases, leveraging its anti-inflammatory and anti-fibrotic properties, and have conducted Phase II trials targeting lung disorders associated with COVID-19 infection and systemic sclerosis.
In the pharmaceutical field, we will also focus on research and business in the field of anti-aging and longevity, which is expected to see rapid business growth on an international scale. While anti-aging and longevity treatments to date have mostly focused on dietary therapy, exercise therapy, supplements, and health foods, the creation of new medical value, such as senolytic drugs (oral medications that eliminate senescent cells and suppress aging-related diseases without promoting cancer), is necessary. Our plasminogen activator inhibitor (PAI)-1 inhibitor RS5614 is a senolytic drug candidate with the potential to promote anti-aging and longevity through oral administration. This not only realizes our company’s mission of “creating new medical treatments that enable people to enjoy lifelong health, both physically and mentally,” but also solves the major social and medical challenge of super-aging, significantly contributing to the enhancement of our corporate value. International recognition and track record are essential, and if our participation in and winning the XPRIZE Healthspan competition enhances the success and reputation of this oral medication as an anti-aging and longevity drug, we expect to expand our business internationally. There is also the example of RS5441 (currently being developed by our licensee, Eirion, as a topical medication for male pattern baldness and age-related alopecia), and we plan to proceed with non-clinical trials of the PAI-1 inhibitor RS5614 for use in the sensory organs (dermatology, etc.) and bone and muscle fields as prescription and over-the-counter drugs.
Open Innovation
By utilizing public funds and resources from external research and medical institutions, we have reduced development costs and implemented highly efficient development. We are considering building multiple value chains based on alliances with external organizations, and our strategies, R&D, and human resource management differ from those of existing startup companies. In fact, we have expanded our pipeline and developed modalities with limited human resources and expenses, and are steadily achieving results. Rather than relying solely on our own resources and internal environment, we actively utilize external resources and external environments to build a framework for efficiently creating innovation. We will promote open innovation and efficient development through collaboration and cooperation with universities and various companies from different industries. Specifically, these include the establishment of open innovation hubs, such as the “Tohoku University x Renascience Open innovation Labo: TREx ” with Tohoku University, the “Hiroshima University x Renascience Open innovation Labo: HiREx ” with Hiroshima University, and the Japan Laboratory at Northwestern University’s Potocsnak Longevity Institute, as well as collaborations with Taipei Medical University and Saudi Arabia’s King Abdullah International Medical Research Center (KAIMRC).
Emphasis on basic research
In order to expand the clinical applications of our seeds, we need to conduct basic research widely and deeply. We strive to discover new clinical applications by providing our compounds as ‘open resources’ to basic researchers internationally for their research. From these, we select appropriate indications from scientific, medical, and commercial perspectives and verify these therapeutic concepts through investigator-initiated clinical trials. We apply for patents for the results of our basic research in collaboration with universities and other research institutions that conducted the collaborative research, striving to secure the intellectual property that forms the foundation of our business. After receiving exclusive licensing rights, we proceed with commercial development.
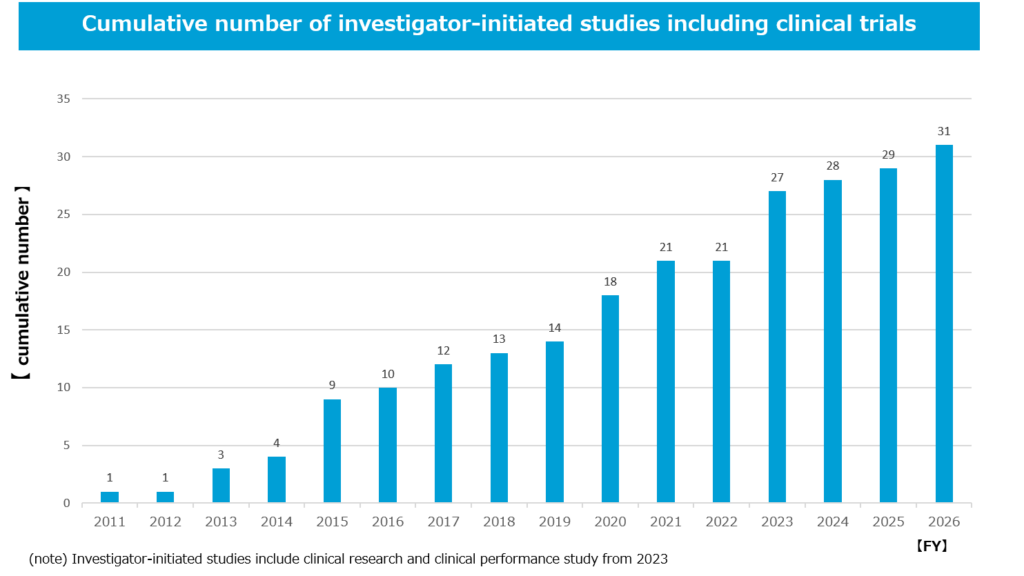
Investigator-initiated clinical trial
We place importance on collaborative research with physician-scientists who conduct a wide range of research, from basic research to clinical trials. Many of the researchers we collaborate with in the field of basic research are also physicians, and are able to conduct investigator-initiated clinical trials themselves as clinical trial coordinators. Since the same researchers often conduct both basic and clinical research, we can efficiently develop a seamless process from basic research to investigator-initiated clinical trials. Our clinical trials are primarily conducted as investigator-initiated trials. We have a track record of 31 investigator-initiated clinical trials to date, and investigator-initiated clinical trials offer many advantages. Because physicians can plan and conduct clinical trials themselves, we can implement them using trial plans and frameworks that suit the challenges and realities of the medical field. As a startup company with limited in-house resources, we have been able to enroll patients in clinical trials for rare and intractable diseases with small patient numbers, such as melanoma, chronic myeloid leukemia, angiosarcoma, and systemic sclerosis, in a relatively short period of time, and also to swiftly conduct clinical trials for COVID-19 pneumonia during the pandemic emergency, as well as to expand into clinical trials for many other cancers, including lung cancer and pancreatic cancer, all thanks to our use of investigator-initiated clinical trials.