IR Information
FAQ
Company Profile
- What is the origin of the company name?
-
Our founder is a nephrologist, and when we first started, there were very few drugs to treat kidney disease. Our company was founded with the goal of developing drugs to treat kidney disease, so we named it Renascience, which means “science of kidneys.” Renascience also has the meaning of “renascence.”
Drug development is not always being done in areas where it is needed, and drug development is lagging behind in areas where it is difficult to develop, such as kidney disease (where efficacy evaluation is difficult to carry out because it takes time) and rare diseases (diseases with few patients and low sales). Therefore, we are currently actively working on drug development in areas where it is difficult to develop and rare diseases.
- What are the reasons for developing various modalities (therapeutic technologies)?
-
Our goal is to create new medical treatments that will solve medical issues and allow people to enjoy lifelong health, both physically and mentally. We are not a venture specializing in a specific technology, so we are not concerned with modality.
The pharmaceutical industry is diversifying from development centered on small molecule drugs to biopharmaceuticals. Furthermore, with recent advances in engineering and information technology, the search for new medical treatments that combine information and engineering technology is also progressing, and major pharmaceutical companies in Europe, the United States, and Japan have already shifted from pharmaceutical-only businesses to businesses that cover medical solutions in general. Medical treatment options are also expanding, including pharmaceuticals, medical devices, and even program medical devices and apps that utilize artificial intelligence (AI).
To that end, in addition to the chemical and biological research that has been our main focus thus far, we would like to broaden our horizons to include engineering and information research, creating a diverse and attractive research and business portfolio.
- What medical issues are you working on?
-
The World Health Organization (WHO) defines important diseases associated with aging and lifestyle habits (aging-related diseases) as “non-communicable diseases (NCDs),” and targets four diseases: cancer, diabetes, respiratory diseases, and cardiovascular diseases. 74% of deaths worldwide in 2023 will be due to these diseases. Our development projects target all four diseases.
In addition, the declining birthrate is also an important social issue, but there are not many pharmaceutical companies that are focusing on this issue. We are also focusing on medical issues such as diseases specific to women and pediatric diseases.
- What are your reasons for emphasizing collaboration with outside research and medical institutions?
-
The role of public research institutes such as universities and medical institutions in creating medical innovation is expanding. Unlike traditional low-molecular-weight synthetic drugs, the technological foundation and seeds of biopharmaceuticals using genetic engineering are held by public research institutes such as universities. In addition, medical institutions own the medical data necessary for developing programmed medical devices using artificial intelligence (AI).
Because we conduct investigator-initiated clinical trials with multiple medical departments at many medical institutions, we have many opportunities to understand medical issues in the field, and we are in an environment where it is easy to obtain the medical data necessary for AI development in a relatively short period of time. Rather than being fixated on our own resources and research environment, we would like to focus on actively utilizing external resources and external environments and build a framework for efficiently creating innovation.
- What is the reason why you can develop the products all the way from basic research to investigator-initiated clinical trials?
-
We place importance on collaborative research with physicians (called physician-scientists) who conduct a wide range of research from basic research to clinical trials. Many researchers who conduct collaborative research in the field of basic research are also physicians, and are able to conduct investigator-initiated clinical trials as clinical trial coordinators (clinical trial directors). In many cases, the same researchers conduct basic research and clinical research, allowing for efficient development through a one-stop process from basic research to investigator-initiated clinical trials.
Furthermore, by developing the drug in-house to the point of close to approval, we are able to ensure that important seeds can be implemented in society, and by deriving it at a later stage of development, we receive larger compensation, which has the advantage of allowing us to return this to our shareholders.
- What are the advantages of the open innovation?
-
Until now, many pharmaceutical companies and drug discovery ventures have focused on building their own pipeline value chains (the accumulation of all development processes) and increasing business value. However, in fields such as pharmaceuticals, where the success rate is extremely low, development periods are long, and investments are large, R&D and business risks are high, so it is essential to form a portfolio that combines many pipelines and diversify risks.
Large pharmaceutical companies have ample funds and are therefore able to develop their own pipeline value chains within existing frameworks. However, this is difficult for companies that do not have abundant funds, such as start-ups.
We have been utilizing the resources of external organizations (research institutes, medical institutions) to improve development efficiency, including costs. We are considering building many value chains based on alliances with external organizations, and our strategies, research and development, and human resource management are different from existing ventures. With few human resources and expenses, we have expanded many pipelines, developed modalities, and are beginning to see results.
Rather than relying solely on our own resources and internal environment, we would like to focus on proactively utilizing external resources and environments to build a framework for efficiently creating innovation. We promote open innovation through collaboration and cooperation with universities and various companies in different industries, and carry out efficient development.
- Why did you establish the Tohoku University Renascience Open Innovation Labo (TREx) and Hiroshima University Renascience Open Innovation Labo (HiREx)?
-
In January 2022, we opened the Tohoku University Renascience Open Innovation Lab (Tohoku University x Renascience = TREx) at the Medicinal Hub of the Tohoku University Graduate School of Medicine (2-1 Seiryou-cho, Aoba-ku, Sendai, Miyagi Prefecture, Faculty of Medicine Building No. 5).
When our company was founded, we had a research institute in the Kawasaki Bio Special Zone in Kanagawa Prefecture, which included a facility for breeding animal models of kidney disease. Later, as our research subjects expanded from kidney disease to many other disease areas and the research stage progressed from basic research to clinical trials, we closed our original research institute, which focused on animal models of kidney disease.
However, we felt that there was a need for a place where cutting-edge scientific and technological achievements could be utilized in many disease areas, a place for face-to-face interaction with doctors and researchers, and a place for open innovation with government agencies and medical industry companies, and so we decided to open TREx.
TREx is also the first base location based on the Agreement on Regional Economic Development between Sendai City and Tohoku University, which was concluded in April 2021.
At TREx, 1) collaboration with different industries such as researchers at the Tohoku University Graduate School of Medicine, doctors at Tohoku University Hospital, companies participating in the Medicinal Hub, and government agencies has been accelerated, 2) research in the existing development pipeline has been promoted and multiple new seeds have been introduced, 3) investigator-initiated clinical trials, medical data acquisition, public funding, and approval strategy planning can be carried out efficiently and quickly, and 4) it has also led to the development and retention of human resources. This has allowed us to further accelerate the high efficiency of research and development, which is one of our strengths.
In April 2023, we signed a comprehensive collaboration agreement with Hiroshima University, and following TREx, we opened the Hiroshima University Renascience Open Innovation Lab (Hiroshima University x Renascience = HiREx). Using HiREx, we are conducting multiple clinical trials, including investigator-initiated clinical trials of pharmaceuticals for non-small cell lung cancer and cutaneous angiosarcoma, and clinical performance tests of programmable medical devices such as AI to support diabetes treatment and AI to support maintenance hemodialysis.
- Why do you place importance on obtaining public research funding?
-
Pharmaceutical research and development, especially the implementation of clinical trials, requires a large amount of research and development costs. Our company’s basic business model is to develop development seeds while utilizing investigator-initiated clinical trials and then license them out to pharmaceutical companies, so we have to bear a large amount of research and development costs ourselves. However, by proactively utilizing public research subsidies (competitive funds), we are able to reduce our own burden of research and development costs required for these high-risk investigator-initiated clinical trials. We would like to continue to proactively obtain and utilize public research subsidies in the future.
Presentation/Outside Activities
- Does Renascience do any social activities?
-
The declining birthrate issue is an important social issue. We are also focusing on medical issues for women (menopausal disorders, AI diagnosis of breast cancer) and children (rapid diagnosis of phenylketonuria). Therefore, we consider it our corporate social responsibility (CSR) to support social activities for children and women. In addition to the specific examples below, we will continue to support awareness-raising activities for children and women.
- Please tell us about your latest academic activities.
-
□ Lecture on “Creating Medical Innovation: Expanding the Role of Academia and Changing Research Environments”
・May 27, 2025: Sapporo Medical University 1st Startup Seminar
At the 1st Sapporo Medical University Startup Seminar (date: May 27, 2025, venue: Sapporo Medical University), Professor Toshio Miyata of the Tohoku University Graduate School of Medicine (Chairman of the Board of Directors of our company) gave a lecture on the theme of “Toward creating medical innovation: The expanding role of academia and changes in the research environment.”
With the recent development of science and technology, the role of academia is expanding. These include biopharmaceutical development, artificial intelligence using medical big data, investigator-initiated clinical trials, and even the launch of startups for practical application. We hope that this seminar will provide an opportunity to consider the research environment required at universities for these initiatives.
□ Lecture on the “Sendai City x Tohoku University Smart Frontier Council Wellness Subcommittee”
・May 29, 2025: Sendai City x Tohoku University Smart Frontier Council Wellness Subcommittee Seminar
At the Sendai City x Tohoku University Smart Frontier Council Wellness Subcommittee (date: May 29, 2025, venue: Tohoku University Graduate School of Medicine (Medicinal Hub)), Professor Toshio Miyata of the Tohoku University Graduate School of Medicine (our Chairman) gave a lecture on cutting-edge longevity research (pharmaceuticals and more) regarding how the Sendai City x Tohoku University Smart Frontier Council Wellness Subcommittee will work together with academia, industry, and government to confront the era of super-aging.
□ Special lecture at the 70th Annual Meeting of the Japanese Society for Dialysis Therapy
・Special lecture at the 70th Annual Meeting of the Japanese Society for Dialysis Therapy, June 27-29, 2025
At the 70th Annual Scientific Meeting and General Assembly of the Japanese Society for Dialysis Therapy (held June 27-29 at the Osaka International Convention Center), Professor Toshio Miyata of the Tohoku University Graduate School of Medicine (CEO of our company, Chairman and President) gave a lecture on the various possible clinical applications of the PAI-1 inhibitor RS5614, which is currently under development, and on the latest research results regarding programmed medical devices that utilize artificial intelligence (AI) to support safe and secure maintenance hemodialysis.
Development Pipeline
- What is important to increase the probability of success in drug development?
-
The important thing in drug development is to confirm safety and efficacy. Safety is carried out according to tests prescribed by pharmaceutical regulations, such as general toxicity and genotoxicity, so it can be done if there is time and funds. On the other hand, evaluating efficacy is not simple, and finding out which diseases a drug is effective against is a difficult task.
Developing a drug takes a lot of time and money. Even if it is not effective for the disease originally envisioned, it may be effective for another disease, so examining the efficacy of the drug for many diseases is important in increasing the probability of success (avoiding failure). It is difficult to broadly examine the indications of this drug (drug repositioning), and it is not realistic to examine it for all diseases.
We provide our developed compounds as “open resources” to researchers affiliated with public research institutes in Japan and overseas. Our framework, which allows us to collaborate with researchers in various fields engaged in cutting-edge basic research, allows us to conduct drug repositioning research far more efficiently and extensively than if we were to conduct basic research using only our own limited resources.
- What are the advantages of investigator-initiated clinical trials?
-
Our clinical trials are investigator-initiated trials conducted by physician scientists who are both researchers and doctors. The overwhelming advantages of investigator-initiated trials are their “quality” and “speed,” in other words, their “efficiency.”
In investigator-initiated clinical trials, doctors who are at the forefront of research and have access to the latest research results, and who treat patients on a daily basis in the medical field, can plan appropriate patient subjects and trial plans.
In addition, because doctors themselves can conduct clinical trials, this is a suitable framework for early-stage clinical trials of unapproved drugs (the stage where usefulness and safety are first confirmed, known as exploratory clinical trials). In addition, development of treatments for orphan diseases (rare diseases. Since the number of patients is small, not much sales can be expected) is not initiated by pharmaceutical companies due to low profitability, so in some cases, it is necessary to conduct investigator-initiated clinical trials from start to finish.
The vast majority of research and development costs are spent at the clinical development stage, not at the basic research stage. Investigator-initiated clinical trials can reduce development costs by quickly utilizing cutting-edge scientific and technological achievements from universities and other institutions, and by allowing clinical trial investigators to appropriately select patients who will be treated.
If we can find an appropriate coordinating physician and obtain support from multiple large medical institutions such as universities, investigator-initiated clinical trials have a significant advantage over corporate clinical trials, and large-scale clinical trials can be conducted in a short period of time, so unlike other companies, we prioritize this type of clinical trial.
- Aren't investigator-initiated clinical trials for the expansion of indications for drugs approved overseas (but not yet approved in Japan) or existing drugs?
-
The 2003 revision of the Pharmaceutical Affairs Law opened the way for investigator-initiated clinical trials, in which doctors themselves conduct clinical trials. However, it remains difficult for doctors to prepare all of the pharmaceuticals required for clinical trials, including safety tests and formulations.
At the time the law was revised, there were many new drugs approved overseas but not approved in Japan, and drugs used off-label (the so-called drug lag), so the main focus of investigator-initiated clinical trials was expanding the indications of drugs unapproved in Japan and drugs used off-label.
Given the ease of conducting clinical trials (which can be done using existing data from manufacturing to safety testing), doctors from many medical institutions, including universities, have been conducting investigator-led clinical trials of new drugs approved overseas (but not approved in Japan) and drugs for off-label use.
There have also been cases where investigator-initiated clinical trials were conducted using existing drugs for rare diseases that pharmaceutical companies are not working on. Because of this background, I think there is still a strong impression that investigator-initiated clinical trials are for drugs approved overseas (not approved in Japan) or for expanding the indications of existing drugs.
However, all clinical trials we conduct are for unapproved drugs (first-in-human), not for drugs approved overseas (not approved in Japan) or for expanding the indications of existing drugs. Our pharmaceuticals are unapproved drugs and we have secured intellectual property rights, so we can commercialize them exclusively and make a sufficient profit.
- Is it possible to apply for regulatory approval with the results of investigator-initiated clinical trials?
-
In our pharmaceutical development, non-clinical trials are conducted in compliance with GLP (Good Laboratory Practice, standards for the implementation of pharmaceutical safety), and investigational drug manufacturing is conducted in compliance with GMP (Good Manufacturing Practice, standards for the manufacturing and quality control of investigational drugs).
In addition, investigator-initiated clinical trials, like corporate clinical trials, are conducted in compliance with Good Clinical Practice (GCP), which is a standard for conducting clinical trials of pharmaceuticals. Therefore, they can be used to apply for approval or obtain licenses.
- What are the reasons for the large number of drugs in your pipeline?
-
We provide the compounds we have developed as open resources to researchers affiliated with universities and other public research institutes both in Japan and overseas. By efficiently conducting a process known as drug repositioning to broaden the scope of indications for our drugs, we have succeeded in increasing our pipeline of numerous pharmaceutical products.
Compounds that are not candidates for clinical development are provided under a material transfer agreement (MTA), and clinical candidate compounds are provided under a collaborative research agreement (a contract that covers research licensing, intellectual property handling, and compensation after commercialization). Regarding our projects, the number of research institutions that have conducted research using compounds provided under MTA as open resources has reached over 100 since our founding, and these have been published in scientific papers alone. Unexpected seeds (concepts) have been born from these many domestic and international collaborative research projects.
- What kind of criteria do you have when deciding on pipeline development?
-
We make pipeline development decisions by comprehensively taking into consideration the four factors listed in the table below: “scientific viability,” “medical viability,” “economic viability (business viability),” and “time frame.”

- What are your considerations regarding intellectual property?
-
Since we conduct joint research with universities and other public research institutes, intellectual property is generally filed jointly with the universities, etc. In addition to complying with the regulations on inventions made during employment at universities, etc., we are required to hold repeated discussions with the university’s industry-academia collaboration department (intellectual property department) and to file, manage, and operate intellectual property in accordance with the university’s rules.
In addition, intellectual property applications are filed upon agreement on the inventors, applicants, ownership ratios of rights, cost burden, and even license conditions (scope, compensation), etc. These negotiations and agreements take a considerable amount of time (several months), so it is necessary to formulate an intellectual property application plan that takes this period into account.
Our intellectual property needs to be covered in Japan as well as in many other countries where we anticipate doing business. We also apply for, maintain, and manage multiple related intellectual property rights, such as substance patents, application patents, and dosage and administration patents, appropriately and continuously.
- Why are you able to conduct many collaborative projects with university researchers?
-
We provide our developed compounds as open resources to researchers affiliated with public research institutes in Japan and overseas. The laboratories of researchers affiliated with public research institutes have research facilities in their specialized fields and their own animal models of diseases, but they are researchers specializing in medicine and biology and do not have any drug candidate compounds.
Due to our open resource policy, we are receiving more and more requests from domestic and overseas researchers who have obtained information about this compound. After filing the patent application, our collaborators are free to present at academic conferences and in papers, and we also allow them to apply for scientific research grants based on our compounds. Our framework, which allows us to develop our products in collaboration with researchers conducting cutting-edge basic research, allows us to conduct research that is far more efficient and comprehensive than basic research conducted with only our own limited resources.
- What are the advantages in research and development of software as a medical device (SaMD) utilizing artificial intelligence (AI)?
-
The application of artificial intelligence (AI) to the medical field is a topic with great potential, but each of the stakeholders who play an important role in research and development is currently facing their own challenges.
Although doctors (medical institutions) are familiar with medical issues and problems (needs) and have a wealth of medical data and ideas, they lack AI methods and networks with IT vendors, and are therefore unable to concretely begin research and development.
On the other hand, although IT vendors with AI technology are interested in applying it to the promising medical field, it is not easy to put it into practical use due to the limited network with doctors (medical institutions) making it difficult to access medical needs and medical data, and the lack of experience in pharmaceutical administration, such as the Pharmaceuticals and Medical Devices Act.
Furthermore, pharmaceutical and health tech companies that want to commercialize the medical application of AI will often find it difficult to handle everything from research to business development on their own, both in terms of time and resources.
Therefore, it is important to have a framework in place from the beginning where doctors (medical institutions) with issues, IT vendors with AI technology, and pharmaceutical and health tech companies with the exit can work together to advance development. Because the product life cycle of programmed medical devices using AI is not as long as that of pharmaceuticals, efficient research and development requires planning from the early stages of development with an eye on approval and licensing and the exit into actual clinical practice.
To achieve this, open innovation across different fields is important, and in addition to doctors, data scientists, AI researchers, and pharmaceutical experts need to work together.
In the course of conducting many investigator-initiated clinical trials, we have built a network with numerous medical institutions and multiple medical departments. This gives us advantages such as easy access to medical issues and medical data (medical support), the ability to conclude joint research project agreements with multiple IT companies through open innovation (technical support), and the ability to comply with pharmaceutical regulations in the process of conducting investigator-initiated clinical trials for pharmaceuticals.
- What are the important aspects of medical applications of artificial intelligence (AI)?
-
The essential elements for research and development of medical AI are 1) medical issues, 2) medical data (quality and quantity), and 3) AI algorithms (engines).Even if there is excellent technology, there are many cases where it is difficult to put it into practical use because it does not match the needs of the medical field or there are issues with using it in the medical field, and this is a problem that many companies with technology face.
In recent years, a technique called “biodesign” has been gaining attention. It involves developing solutions based on the needs of medical professionals and then optimizing them while imagining the final product in the clinical setting. The same is true for programmed medical devices with AI as their core technology. While AI technology is certainly important, the medical data it develops is not targeted at machines but at patients, each with their own unique characteristics. Customizing AI based on medical issues, medical data, and the advice of medical professionals is crucial.
Medical data is an essential resource for AI research. While AI uncovers patterns from large amounts of data (inductive inference), a sufficient amount of data is undoubtedly necessary, data quality is just as important. Furthermore, the optimal AI algorithm must be selected based on the medical problem being solved and the type of medical data being utilized. Rather than searching for a medical field where a specific AI algorithm can be applied, it is necessary to “select (or, in some cases, develop) the optimal AI algorithm” to solve a specific medical problem. Furthermore, once an AI algorithm is selected, data scientists can use the data to analyze it, but only doctors can interpret and determine whether the results are correct. Therefore, without the involvement of doctors, it is impossible to train AI on high-quality medical data and solve problems.
Collaboration between doctors, data scientists, and AI researchers is important, and the active involvement of medical professionals (doctors) who have the medical issues, medical data, experience, and knowledge is key to successfully using AI to solve medical problems.
To promote research and development into the medical application of artificial intelligence, we signed a memorandum of understanding with NEC Solution Innovators, Ltd. in November 2022 and a joint research agreement (Memorandum of Understanding) with NEC Corporation in June 2023.
- How many pipelines are appropriate for the size of the company?
-
By providing our developed compounds as open resources to researchers affiliated with universities and other public research institutions both in Japan and overseas, we are able to efficiently explore the possibility of expanding the range of indications for our drugs within our limited resources (a process known as drug repositioning).
Because we do not require any in-house resources, there are no restrictions on the number of non-clinical trials (tests using disease animal models). Clinical development is conducted as investigator-initiated trials, utilizing contract research organizations (CROs), which reduces our in-house human resources.
We are steadily progressing with nine clinical trials conducted in fiscal 2023, 10 in fiscal 2024, and nine planned for fiscal 2025 (including one clinical performance study). Our goal is to continue conducting at least five investigator-initiated clinical trials per year.
- What pipeline is currently a high priority?
-
Pipeline development that is advanced in clinical development is a high priority.
In the pharmaceutical field, we are focusing on the development of plasminogen activator inhibitor-1 (PAI-1) inhibitors in the oncology and respiratory fields. In the oncology field, Phase III trials are underway for chronic myeloid leukemia and malignant melanoma, and Phase II trials are underway for non-small cell lung cancer and cutaneous angiosarcoma. In the respiratory field, Phase II trials for COVID-19 lung injury have been completed, and Phase II trials for interstitial lung disease associated with systemic sclerosis are underway.
Research and development of medical devices (ultra-thin endoscopes) is almost complete. The main fiberscope component was approved in December 2022, and development of the accessory guide catheter is almost complete, so we plan to proceed with preparations for approval.
In terms of programmable medical devices utilizing artificial intelligence (AI), development is progressing ahead with AI for diabetes treatment support and AI for maintenance hemodialysis medical care support. Clinical performance testing for approval applications will begin in 2024, and at present, the clinical performance testing has been completed and proof of concept (POC) has been obtained for diabetes and dialysis.
- What is the reason for developing the PAI-1 ihhibitor in respiratory disease?
-
PAI-1 is a protein necessary for the dissolution of blood clots, but is also deeply involved in inflammation and tissue fibrosis. Based on this mechanism, we are developing it for respiratory diseases. Specifically, we are currently conducting projects for COVID-19 lung injury (Phase II trial completed) and interstitial lung disease associated with systemic sclerosis (Phase II trial).
- What is the reason for developing the PAI-1 inhibitor in oncology?
-
High PAI-1 expression in cancers is known to be associated with higher malignancy and poor prognosis (the “PAI-1 paradox”). Through collaborative research, we discovered that PAI-1 promotes the expression of immune checkpoint molecules (such as PD-L1) in cancer cells. In mouse models of colon cancer and melanoma, administration of our PAI-1 inhibitor reduced the expression of immune checkpoint molecules in cancer cells, increased the infiltration of cytotoxic T cells into tumors, and suppressed tumor-associated macrophages.
Based on the results of these non-clinical studies, we conducted a Phase II study in melanoma and confirmed its expected efficacy. Specifically, an eight-week combination of nivolumab and RS5614 in patients with melanoma who were refractory to nivolumab resulted in a response rate of 24.1% (7/29 patients), exceeding the response rate of the existing combination of nivolumab and ipilimumab (the response rate for the combination of nivolumab and ipilimumab in Japan was 13.5%). Based on this concept, we are also conducting a phase II investigator-initiated clinical trial targeting non-small cell lung cancer, confirming that the PAI-1 paradox is indeed involved in cancer treatment and that the combination of PAI-1 inhibitors is effective in some cancer types.
- How was the PAI-1 inhibitor obtained?
-
Based on the crystal structure of the human PAI-1 molecule, we used computational engineering to search a virtual compound library of approximately 2 million compounds and identified 96 PAI-1 inhibitor candidate compounds. Using PAI-1 inhibitory activity as an indicator, we have synthesized over 1,400 new inhibitor compounds over the course of more than 10 years. After screening for efficacy and evaluating their safety and efficacy, we identified RS5275, an orally bioavailable lead compound with excellent safety. Further synthesis of RS5275 yielded four clinical candidate compounds: RS5441, RS5484, RS5509, and RS5614. RS5614 was ultimately selected as the candidate compound for clinical development.
- How is the status of domestic and international competition for PAI-1 inhibitor development?
-
In the past, many pharmaceutical companies and biotech ventures, including major ones both in Japan and overseas, have worked on developing small molecule PAI-1 inhibitors. Some drugs have been reported to be effective in animal models of mice and rats, and PAI-749 (Diaplasinin) even made it to the clinical stage. However, no effect was observed in humans, and development was discontinued after Phase I clinical trials.
- Are there any safety issues with the PAI-1 inhibitor?
-
Non-clinical studies have included safety pharmacology studies (hERG studies, rat central nervous system studies, monkey cardiovascular and respiratory system studies), general toxicity studies (a 26-week oral administration study in rats, and a 39-week oral administration study in monkeys), genotoxicity studies, phototoxicity studies, and reproductive and developmental toxicity studies to confirm that there are no issues for clinical trials or regulatory approval applications.
A Phase I single-dose study confirmed the safety of RS5614 up to 240 mg, and a Phase I repeated-dose study observed mild adverse events following oral administration of 120 mg for seven days. RS5614 has been administered to over 200 subjects (healthy individuals, patients with chronic myeloid leukemia, patients with COVID-19 lung injury, and patients with melanoma, among others). In a 48-week study of patients with chronic myeloid leukemia, 180 mg daily was administered (33 cases). No serious adverse events related to the investigational drug have been reported, and the drug is considered to be highly safe.
- What is the reason for developing the PAI-1 inhibitor in chronic myeloid leukemia (CML)?
-
CML, a type of blood cancer, develops when chromosomal abnormalities occur in hematopoietic stem cells, leading to the uncontrolled proliferation of cancerous leukemia cells (CML cells).
The mainstay of treatment for CML is the tyrosine kinase inhibitor TKI (such as imatinib). However, while TKIs act on CML cells, they do not affect CML stem cells, which reside in bone marrow niches and are the source of CML. Therefore, discontinuing TKI treatment allows CML cells to proliferate, preventing a complete cure of CML. It has been shown that PAI-1 inhibitors act on CML stem cells, enhancing the action of TKIs, thereby leading to a complete cure of CML. In fact, when RS5614 was administered in combination with a TKI in CML model mice, the number of CML stem cells remaining in the bone marrow was significantly reduced compared to administration of the TKI alone, significantly improving survival rates.
Although the development of TKIs has significantly improved the prognosis for CML patients, long-term, expensive TKI treatment is required to cure CML. Side effects from long-term continuous use are also a problem. Therefore, it is important to achieve a cure (treatment-free remission, TFR) that does not require TKI use as soon as possible. RS5614 is expected to be a safe drug with a new mechanism of action that will quickly lead many CML patients to TFR.
- What were the results of the Phase II trial for chronic myeloid leukemia (CML)?
-
In the Phase IIa trial, CML patients who had been receiving TKI treatment for at least two years were administered RS5614 120 mg/day for four weeks (TKI treatment continued for 12 weeks). The investigator-initiated trial was conducted at Tohoku University, Akita University, and Tokai University, and the rate of DMR (deep molecular response: highest therapeutic effect) achieved after 12 weeks was used as an indicator. 21 patients were enrolled, and there were no dropouts or discontinuations, so all were included in the analysis. The primary endpoint was that four of the 21 patients achieved DMR, with a cumulative DMR rate at 12 weeks of 20.0% (the average cumulative DMR rate at three months with previous TKI monotherapy was 2.0% (treatment with TKI alone was referred to as the historical control)). In the safety evaluation, no side effects were observed in any of the 21 patients included in the analysis.
In the Phase IIb study, CML patients were administered a combination of a TKI and RS5614 (starting at 150 mg/day, with the option to increase to 180 mg/day), with the objectives of confirming that a cumulative DMR achievement rate of 33.0% could be expected 48 weeks after the start of RS5614 administration, compared to 8.0% in historical controls, and of confirming the pharmacokinetics and safety of RS5614 when administered long-term in combination with a TKI. Of the 33 patients, 11 achieved a DMR, with a cumulative DMR achievement rate of 33.3% at 48 weeks (a result equal to or better than expected (POC)). Regarding safety, there were no serious adverse events causally related to the investigational drug.
- Please tell us about the Phase III trial for chronic myeloid leukemia (CML).
-
Based on the results of the late Phase II study, a placebo-controlled, double-blind, investigator-initiated Phase III clinical trial to evaluate the efficacy of the combination of TKI and RS5614 in patients with chronic phase CML is underway in collaboration with 12 universities and medical institutions, including Tohoku University, Tokai University, and Akita University. This study was adopted by the Japan Agency for Medical Research and Development (AMED) under the Research Program of the “Practical Application for Innovative Cancer Therapy” in fiscal 2022 (Tohoku University is the representative research organization and Renascience participates as a sharing research organization). In November and December 2021, we held face-to-face consultations with the Pharmaceuticals and Medical Devices Agency (PMDA), and in May 2022, we submitted a notification of the clinical trial plan to the PMDA and started a multicenter Phase III study. Sixty (60) patients with chronic stage CML who had been treated with TKI for more than 3 years will be enrolled in the study, and the study will evaluate whether the combination of RS5614 significantly increases the maintenance rate of DMR for more than 2 years compared to TKI alone in 60 patients with chronic CML who have been treated with TKI for more than 3 years.
- What is the reason for developing the PAI-1 inhibitor in malignant melanoma?
-
Currently, there are four major cancer treatments: 1) surgery, 2) radiation therapy, 3) chemotherapy (anticancer drugs), and 4) immunotherapy. Of these, immunotherapy utilizes the body’s natural immune system to attack cancer. Various immunotherapies have been proposed, but the most effective are immune checkpoint inhibitors, which inhibit the immune brakes. Because excessive immune responses are harmful, the body has mechanisms to suppress them. The molecules that perform this brake function are called immune checkpoint molecules. In fact, cancer exploits these immune checkpoint molecules to prevent its own immune system from working against itself.
Immune checkpoint inhibitors inhibit immune checkpoint molecules, thereby releasing these brakes and activating the immune response against cancer. We have discovered that PAI-1 inhibits cancer immunity via immune checkpoint molecules. In animal model studies, it was found that cancers such as malignant melanoma and colon cancer regressed in animals administered RS5614, and that when administered in combination with an immune checkpoint inhibitor antibody, it strongly synergistically enhanced cancer immunity.
The immune checkpoint inhibitory effects of the PAI-1 inhibitor RS5614 were confirmed in a Phase II trial of malignant melanoma.
(What is malignant melanoma?)
Malignant melanoma is a type of skin cancer that develops when melanocytes, the skin cells that produce melanin, the pigment responsible for skin color, become malignant. It has a high metastasis rate and is considered highly malignant among skin cancers. While the number of melanoma patients in Japan is low, at 1.5-2 per 100,000, the rate in the United States is 21.0, several dozen times higher than in Japan. Malignant melanoma is an extremely malignant cancer (the 5-year survival rate is approximately 50% for tumors over 4 mm in size, approximately 40% for those with regional lymph node metastasis, and several percent for those with distant metastasis). Furthermore, it has been reported that the progression rate of malignant melanoma in Japan is approximately three times higher than in the United States. This is because malignant melanoma in Japan is often of the acral lentigo type, which differs from that in Europe and the United States and is thought to make treatment less effective.
Treatment for malignant melanoma begins with surgical removal. However, in many cases, cancer is already advanced at the time of detection, and in cases where radical resection is not possible, chemotherapy (drugs) is required. Radiation therapy is not very effective for malignant melanoma. The introduction of antibody drugs (immune checkpoint inhibitors) such as nivolumab that target immune checkpoint molecules has led to groundbreaking advances in chemotherapy. However, the response rate of nivolumab for malignant melanoma in Japan is only 22.2%. Furthermore, for patients in whom nivolumab is ineffective, combination therapy with nivolumab and ipilimumab has been approved, but the response rate is 21% overseas and 13.5% in Japan. Nivolumab-ipilimumab combination therapy causes severe side effects in over half of patients, and the incidence of severe immune-related side effects leading to treatment discontinuation is four times higher than with monotherapy, resulting in months of hospitalization and interruption of cancer treatment. Furthermore, given the high medical costs, there is a strong need for a combination drug that uses a different modality from antibodies, can be administered orally, has fewer side effects, and increases response rates.
- Please tell us the results of the Phase II trial for malignant melanoma.
-
A Phase II investigator-initiated clinical trial was conducted in collaboration with six medical institutions, including Tohoku University Hospital, to investigate the efficacy and safety of combining the PAI-1 inhibitor RS5614 with the immune checkpoint inhibitor nivolumab in patients with malignant melanoma that is difficult to completely resect surgically. The results showed that the combination of nivolumab and RS5614 was equally or more effective than the reported combination of nivolumab and ipilimumab, an existing treatment (response rate of 24.1% for nivolumab + RS5614, 13.5% for nivolumab + ipilimumab). Furthermore, while there are safety concerns regarding the combination of nivolumab and ipilimumab due to serious immune-related side effects, no problematic side effects were observed with the combination of nivolumab and RS5614.
Results of a Phase II Investigator-Initiated Clinical Trial
Efficacy
・The primary efficacy endpoint, the response rate after 8 weeks of concomitant use of RS5614, was 24.1%.
・The disease control rate (complete response (CR) + partial response (PR) + stable disease (SD)) was 62.0%.
Safety
・Of the 34 patients included in the safety analysis, 11 serious adverse events occurred in 9 patients by 8 weeks of treatment, with 2 cases of liver dysfunction (5.9%) possibly related to the study drug.
- How will RS5614 change the treatment of malignant melanoma?
-
Currently, in melanoma, insurance coverage is available for the combination of nivolumab and ipilimumab in patients who are resistant to nivolumab, but serious side effects associated with the nivolumab-ipilimumab combination have become a problem. The Japan Clinical Oncology Group (JCOG) began a Phase III trial (JCOG2007 trial) at 59 facilities nationwide in April 2021 to compare the efficacy of nivolumab + ipilimumab combination therapy in patients with non-small cell lung cancer. However, the trial was discontinued on March 30, 2023, due to an unexpectedly high treatment-related death rate of approximately 7.4% (11 out of 148 patients) in the nivolumab-ipilimumab combination group. Thus, the combination of nivolumab and ipilimumab presents problems regardless of the type of cancer.
In a Phase II trial targeting malignant melanoma, the combination of RS5614 and nivolumab demonstrated equal or greater efficacy than the combination of nivolumab and ipilimumab in patients for whom nivolumab was ineffective, and the combination of nivolumab and RS5614 demonstrated greater safety than the combination of nivolumab and ipilimumab. Based on the above, the development of a new treatment for unresectable malignant melanoma for which immune checkpoint inhibitor monotherapy is ineffective is an urgent issue, and the combination of nivolumab and RS5614 is expected to be a drug therapy with high efficacy and safety.
- Is RS5614 useful in the treatment of other solid tumors?
-
Non-clinical studies using animal models have shown that oral administration of the PAI-1 inhibitor RS5614 can cause regression of not only malignant melanoma, but also other cancers such as colorectal cancer and lung cancer, and that this effect is significantly enhanced when combined with immune checkpoint inhibitor antibodies. Therefore, we have begun a Phase II investigator-initiated clinical trial for non-small cell lung cancer and a Phase II investigator-initiated clinical trial for cutaneous angiosarcoma in collaboration with Hiroshima University Hospital and other medical institutions.
- What is the medical significance of the clinical trial for malignant melanoma?
-
It has previously been reported that cancers with high PAI-1 expression are highly malignant and have poor prognoses, a phenomenon known as the “PAI-1 paradox.” Through collaborative research with numerous universities both in Japan and abroad, we have discovered that cancer cells evade attack from the immune system by producing PAI-1 and enhancing the expression of immune checkpoint molecules such as PD-L1. In preclinical studies using animal models, we have demonstrated that oral administration of the PAI-1 inhibitor RS5614 can cause regression of melanoma, colon cancer, lung cancer, and other cancers. In this clinical trial, we administered RS5614 for eight weeks in combination with 29 melanoma patients for whom surgical resection was difficult and for whom nivolumab was ineffective. Seven patients responded to RS5614. RS5614 has already been demonstrated to be effective as a drug therapy for chronic myeloid leukemia, a type of blood cancer, in Phase II trials (early and late phases), and Phase III trials are currently underway. These findings demonstrate that the “PAI-1 paradox” is indeed important in cancer treatment, that PAI-1 is a therapeutic target for some types of cancer, and that PAI-1 inhibitors are effective as drug therapy in humans.
- Please tell me about the Phase III trial for melanoma.
-
Based on the results of the Phase II trial, we are currently conducting a Phase III randomized, placebo-controlled, double-blind, investigator-initiated clinical trial in collaboration with 18 universities and medical institutions, including Tohoku University, Sapporo Medical University, and Kumamoto University, to evaluate the efficacy and safety of combining nivolumab with RS5614 in 124 patients with unresectable melanoma. This trial was selected for the National Institutes of Biomedical Innovation, Health, and Nutrition’s FY2025 “Orphan Drug Research Grant Program.”
The trial was approved by the Institutional Review Board (IRB) at Tohoku University Hospital in January 2025, and the Phase III trial began in February 2025, with the first dose administered. This trial will evaluate whether the combination of nivolumab with the investigational drug RS5614 significantly extends survival compared to the nivolumab monotherapy group in 124 patients with unresectable melanoma.
- When will RS5614 be available for use in the medical field as a treatment for malignant melanoma?
-
We were able to confirm its efficacy in a Phase II trial. We will continue discussions with regulatory authorities and, depending on the results of the Phase III trial currently underway, will clarify our future roadmap, including approval applications. Furthermore, as we do not hold a pharmaceutical manufacturing and sales license, we hope to clarify the path to commercialization through collaboration with other companies.
- What is the difference between RS5614, a PAI-1 inhibitor, and other immune checkpoint inhibitors?
-
All immune checkpoint inhibitors to date are antibody drugs that require hospitalization and administration by injection, making them expensive. Furthermore, existing antibody drugs are known to have various side effects. RS5614 is highly safe and is expected to be an oral medication that can be taken at home. Furthermore, unlike antibodies, it is produced by chemical synthesis, so its price is expected to be lower than that of antibodies.
- What is the reason for developing the PAI-1 inhibitor in lung cancer?
-
Standard treatment for non-small cell lung cancer is platinum-based chemotherapy and immunotherapy using anti-PD-1/PD-L1 antibodies, but few cases are curative. In cases where this treatment is ineffective, chemotherapy such as docetaxel is administered as second-line treatment, but survival times are short at three months, necessitating third-line treatment. Effective third-line treatments are scarce, and new therapeutic agents are eagerly awaited.
Through collaborative research, we have discovered that PAI-1 is involved in the progression and proliferation of lung cancer, and confirmed that cancer cells resistant to anti-PD-1 antibodies express extremely high levels of PAI-1. In fact, in a non-clinical study using a lung cancer model mouse, we confirmed that combined administration of an anti-PD-1 antibody and a PAI-1 inhibitor demonstrated greater anti-cancer activity than administration of an anti-PD-1 antibody alone.
Based on these findings, we are conducting a Phase II trial at Hiroshima University Hospital and other locations to confirm the efficacy and safety of the PAI-1 inhibitor RS5614 in non-small cell lung cancer.
- What is the reason for developing the PAI-1 inhibitor in cutaneous angiosarcoma?
-
Cutaneous angiosarcoma is a rare soft tissue tumor (approximately 300 cases per year in Japan), and apoptosis-inducing taxane anticancer drugs are the first-line treatment. However, a prognosis study of 90 cases of cutaneous angiosarcoma treated with taxane anticancer drugs found that the overall survival rate was 649 days, indicating limited therapeutic benefit. Research and development of new therapeutic agents is urgently needed. PAI-1 is primarily produced in the vascular endothelium.
In our analysis of cutaneous angiosarcoma samples, we found that high PAI-1 expression strongly correlates with disease prognosis. The finding that cancer cells with high PAI-1 expression are resistant to apoptosis strongly suggests that the combined use of a PAI-1 inhibitor may enhance the therapeutic efficacy of taxane anticancer drugs in angiosarcoma. Therefore, a Phase II trial (investigator-initiated clinical trial) is being conducted at Tohoku University and other institutions to confirm the effectiveness of the PAI-1 inhibitor RS5614 in patients with cutaneous angiosarcoma in whom the taxane anticancer drug paclitaxel has failed.
*Apoptosis: A phenomenon in which a cell’s own death program is activated, leading to cell death, as opposed to necrosis , in which cells die due to injury.
- What is the reason for developing the PAI-1 inhibitor in systemic scleroderma?
-
Systemic sclerosis (SSc) (designated intractable disease 51) is a systemic autoimmune disease characterized by vascular damage and fibrosis in the skin and many organs, but its cause remains unknown. It is an intractable disease that causes symptoms such as skin hardening, pulmonary fibrosis (interstitial lung disease, ILD), gastroesophageal reflux disease, cardiac lesions, and digital ulcers. It is estimated that there are more than 30,000 SSc patients in Japan.
ILD is a serious disorder that accounts for more than 30% of deaths among SSc patients. Even when ILD is not the direct cause of death, pulmonary function is impaired due to fibrosis, resulting in severe coughing and dyspnea, significantly limiting daily life. Current treatments are not effective enough, and despite current advances in autoimmune disease treatment, disease-related deaths account for less than 10% of all deaths in autoimmune diseases such as rheumatoid arthritis and systemic lupus erythematosus. However, in SSc, disease-related deaths account for 70% of deaths, creating a strong demand for the development of effective treatments.
RS5614, currently under development by our company, inhibits inflammation, fibrosis, and vascular damage. Because the pathology of SSc is primarily characterized by autoimmune inflammation, fibrosis, and vascular damage, it has been suggested that RS5614 may slow the progression of SSc symptoms. In fact, in an SSc animal model, RS5614 has been demonstrated to be more effective at inhibiting pulmonary fibrosis than existing drugs.
While many current SSc treatments, such as steroids and immunosuppressants, have strong side effects, RS5614’s high safety profile suggests its potential as a treatment for ILD associated with SSc. Therefore, a Phase II trial to investigate the efficacy and safety of the PAI-1 inhibitor RS5614 for SSc-ILD is being conducted at Tohoku University Hospital and other medical institutions in Japan.
This trial has also been selected for the Japan Agency for Medical Research and Development (AMED) fiscal year 2023 “Research Project for the Practical Application of Intractable Diseases” (Tohoku University is the principal research institution, and our company is also participating as a contributing research institution).
- What is the reason for the PAI-1 inhibitor in novel coronavirus lung injury?
-
The global spread of the novel coronavirus disease (COVID-19) is a significant social issue. While approximately 80% of infected individuals experience mild symptoms, the disease can progress to severe pneumonia and acute respiratory distress syndrome (ARDS), particularly in elderly patients and those with underlying medical conditions. While mild cases are treated at home or in an inpatient facility, some patients who initially experience mild symptoms rapidly develop severe symptoms, creating a problem. Vaccination and a mutated virus strain (the Omicron strain) have made the disease less severe than before, but we must continue to monitor future developments, including the possibility of further mutated strains worsening the disease. Furthermore, with the COVID-19 classification being downgraded from “Category 2” to “Category 5” under the Infectious Diseases Control Act, we expect the number of patients receiving treatment at home or in outpatient settings to increase. We are preparing to rapidly conduct next-generation clinical trials in the event of the emergence of new pneumonia-causing strains.
We aim to develop therapeutic drugs to prevent pneumonia from worsening, particularly as safe and convenient preventive and therapeutic drugs (oral medications) that can be prescribed in outpatient clinics and taken at home, thereby contributing not only to extending patients’ lives but also to reducing the burden on medical facilities and making more efficient use of medical resources. Patients with severe pneumonia caused by COVID-19 experience rapid progression of lesions such as inflammation and fibrosis, and characteristic findings of hypercoagulation are observed. Significant findings of COVID-19 lung injury are intrapulmonary fibrin microthrombi, inflammation, and fibrosis. RS5614, which we are developing, inhibits inflammation, fibrosis, and vascular damage, and is therefore thought to be effective in treating COVID-19 lung injury.
- What were the results of the Phase II studies of novel coronavirus lung injury?
-
An early Phase II investigator-initiated clinical trial (open-label) was conducted from the fall of 2020, and the clinical trial report was completed in June 2021. No notable side effects were reported, and all 26 patients who were hospitalized with lung injury and administered the investigational drug were successfully discharged.
Based on the results of the early Phase II investigator-initiated clinical trial, we conducted a placebo-controlled late Phase II investigator-initiated clinical trial in patients with COVID-19 lung injury (moderate, hospitalized) in collaboration with 20 domestic universities and medical institutions, including Tohoku University, Kyoto University, Tokyo Medical and Dental University, and Tokai University. This trial was selected for AMED’s “Project to Promote the Development of Innovative Drugs for Emerging and Re-emerging Infectious Diseases” in March 2021 (Tohoku University as the principal investigator, and our company as a contributing research institution). Following a preliminary interview with the PMDA in April 2021, the study protocol was finalized and began in June 2021. Due to a sharp decline in the number of infected patients and the emergence of the Omicron strain, the number of eligible COVID-19 lung injury patients (moderate, hospitalized) decreased. The clinical trial was extended until the end of October 2022 (75 enrolled cases), and the clinical trial report was completed in April 2023.
While no statistically significant difference was observed between the two groups for the primary efficacy endpoint, the RS5614 group demonstrated a greater suppression of progression compared to the placebo group, suggesting its efficacy, particularly in patients with relatively mild symptoms. Furthermore, the proportion of patients requiring oxygen therapy within 3 to 5 days of hospitalization was lower in the RS5614 group, suggesting the efficacy of RS5614 in early treatment. Furthermore, unlike the placebo group, the RS5614 group also demonstrated improvement in pneumonia imaging findings. The incidence of adverse events was similar between the RS5614 and placebo groups, confirming the safety of administering this investigational drug (RS5614) to patients with COVID-19 lung injury.
- Is the PAI-1 inhibitor effective in preventing aging?
-
In collaboration with Northwestern University in the United States, we have conducted research on “PAI-1 and aging.”
Living cells cannot proliferate indefinitely due to a phenomenon known as cellular senescence. This phenomenon is caused by shortening of gene telomere length and cellular senescence factors such as p53. It is known that senescent cells express extremely high levels of PAI-1 in addition to p53. In fact, it has been shown that cellular senescence can be inhibited by suppressing p53 and PAI-1. It has been reported that PAI-1 expression is high not only in cells, but also in aged tissues and individuals (klotho mice, and humans with Werner syndrome, a well-known premature aging disorder). In klotho mice, a well-known aging model, we have shown that inhibiting PAI-1 expression and activity at the gene or protein level can improve all of the cardinal symptoms of aging.
Furthermore, a study of members of the Amish community, a Christian sect living in the Midwestern United States, found that those without the PAI-1 gene lived 10 years longer than those with it. The researchers also found that those with the gene deficiency were less susceptible to diseases such as diabetes.
These findings were reported in numerous newspapers, including the New York Times in November 2017. The lead researcher, a professor at Northwestern University, stated, “Not only are they living longer, but they are also living healthier lives. They are the ideal longevity model.” This human epidemiological study is consistent with the results of cell and mouse experiments. With aging, diseases such as cancer, vascular (atherosclerosis), lung (emphysema, chronic obstructive pulmonary disease), metabolism (diabetes, obesity), kidney (chronic kidney disease), bone and joint (osteoporosis, osteoarthritis), and brain (cerebrovascular disease, Alzheimer’s disease, dementia) develop. Interestingly, PAI-1 expression is extremely high in tissues associated with these diseases. Furthermore, it has been confirmed that administering PAI-1 inhibitors to animal models of these diseases significantly improves their pathology.
Recently, it has been shown that senescent cells express proteins (immune checkpoint molecules) that act as brakes on the immune system, allowing them to escape immune attack. When aging mice or mice with lifestyle-related diseases are administered drugs that block immune checkpoint molecules (immune checkpoint inhibitors), the immune system is activated, removing senescent cells and improving organ and tissue aging and lifestyle-related diseases. We have discovered that PAI-1 is involved in the expression of immune checkpoint molecules in cancer cells, promoting cancer cell proliferation, and that PAI-1 inhibitors have immune checkpoint inhibitory effects. Thus, PAI-1 promotes cancer and aging, and our PAI-1 inhibitors are expected to be effective against cancer and other aging-related diseases.
In conclusion, fascinating insights are emerging regarding PAI-1 and aging. We are currently planning a specific clinical study funded by Xprize to examine the effects of PAI-1 inhibitors on aging.
- Why develop nucleic acid drugs?
-
Our company is researching and developing medical solutions using various modalities to solve medical challenges, and we are conducting investigator-initiated clinical trials for multiple pipelines. Luxana Biotech, our collaborative research partner in nucleic acid drugs, is researching and developing highly effective and safe biopharmaceuticals (nucleic acid drugs) using a drug discovery platform technology utilizing artificially modified nucleic acids developed by Professor Satoshi Kobiga of the Graduate School of Pharmaceutical Sciences at Osaka University. Therefore, we have decided to jointly conduct research, clinical development, and commercialization of novel nucleic acid drugs, leveraging Luxana Biotech’s artificially modified nucleic acid technology and our track record and experience in investigator-initiated clinical trials.
This research has been selected for the Japan Agency for Medical Research and Development (AMED)’s “Smart Bio Drug Discovery Research Support Project” (Osaka University is the lead research institution, and our company is a contributing research institution) in fiscal year 2024.
- What is SaMD utilizing AI for supporting diabetes treatment?
-
Insulin injection therapy is necessary to strictly control blood sugar levels and prevent diabetic complications. However, the safe dosage range of insulin is narrow, and overdosing can lead to hypoglycemia, so the optimal type and dosage must be selected for each patient. Meanwhile, diabetes specialists account for less than 2% of physicians and are unevenly distributed geographically. Currently, diabetes patients are not always treated by a diabetes specialist; rather, they often consult a non-specialist.
To address this issue, we are collaborating with Tohoku University and NEC Corporation to develop DM-SAiL (SAiL stands for Skill Acquisition Learning), an AI program that helps non-specialists administer specialist-level insulin treatment. We have completed analysis of patient data from approximately 1,000 patients (approximately 1,080,000 clinical parameters) admitted to Tohoku University Hospital, and have developed an AI that can predict insulin dosages prescribed by specialists with an error of approximately 2 units. The accuracy rate for patients at Tohoku University Hospital was approximately 90% for both rapid-acting and long-acting insulin, with an error of 0.65 to 0.95 units from the prescriptions of diabetes specialists, demonstrating extremely high results.
A PMDA protocol consultation was conducted in February 2024, and clinical performance testing for drug approval began in August 2024. In March 2025, data from the target number of 130 cases was obtained and the final analysis of the study results was completed. The analysis resulted in an average accuracy rate of 85.46%, 5% higher than the initial target accuracy rate of 80% for the primary endpoint, demonstrating non-inferiority (equivalent) of AI predictions to specialists.
Clinical Performance Test Results
- The average accuracy rate for the 116 cases analyzed was 85.46, with a confidence interval of (83.59, 87.34).
- The mean absolute error (MAE = |AI prescription units – specialist prescription units) between the insulin dosage units used in specialist treatment and the insulin dosage units recommended by the test device was an average of 1.61.
- As a result, Proof-of-Concept was obtained.
This study has also been selected for the Japan Agency for Medical Research and Development (AMED)’s FY2022 “Medical-Engineering Collaboration Innovation Promotion Project (Development and Commercialization Project) (our company is the principal research institution).”
- What is SaMD utilizing AI for supporting maintenance hemodialysis?
-
Patients with chronic kidney failure undergo hemodialysis three times a week for the rest of their lives to remove fluid and waste products in place of their failed kidneys. While insufficient fluid removal can lead to cardiopulmonary dysfunction, including heart failure and high blood pressure, excessive fluid removal can cause hypotension during dialysis, leading to adverse events such as nausea and loss of consciousness. If inadequate or excessive fluid removal results in adverse events, medical professionals will be overwhelmed and placed under a heavy burden on patient care.
To ensure safe and reliable hemodialysis, we are jointly developing an AI (Dual-Channel Combiner Network, DCCN) that predicts the appropriate target total fluid removal volume with Tohoku University and NEC Corporation (NEC). We have developed an AI system that trains on 725,000 dialysis records (patient information, dialysis information, and test information) obtained from St. Luke’s International Hospital and private dialysis medical facilities. Using the patient’s records from the past five dialysis sessions and pre-dialysis data from the day of dialysis, we can predict the probability of intradialytic blood pressure drop (below 20 mmHg) with a high accuracy of AUC 0.91 before the start of dialysis. Furthermore, we can predict the total fluid removal volume based on the doctor’s empirical setting with an error of about one cup. We completed the PMDA pre-development consultation in April 2023. We are currently working toward the practical application of a programmable medical device centered on this AI.
We conducted a PMDA protocol consultation in January 2024, and began clinical performance testing for pharmaceutical approval in October 2024. In July 2025, we obtained data from the required number of 108 cases and are analyzing the test results. The preliminary analysis results showed a mean accuracy rate of 90.31%, 10% higher than the initial target accuracy rate of 80% for the primary endpoint, demonstrating non-inferiority (equivalent) of the AI’s predictions to specialists.
Clinical Performance Test Results (Preliminary)
- The mean accuracy rate for the 108 analyzed cases was 90.31, with a confidence interval of (89.00, 91.62).
- The mean absolute error (MAE = AI prescription unit – specialist prescription unit) between the target fluid removal volume prescribed by the specialist and the recommended fluid removal volume presented by the test device was 117.9.
- Based on these results, proof of concept is expected.
As part of efforts toward practical application, the company has also begun developing an AI system to be installed in hemodialysis equipment that will control the amount of fluid removal and blood flow during hemodialysis, and signed joint development agreements with Toray Medical Co., Ltd. in December 2023 and Nipro Corporation in March 2024.
This trial has also been selected for the Japan Agency for Medical Research and Development (AMED) fiscal year 2023 “Medical Device Development Promotion Research Project” (with joint research partner Tohoku University as the principal research institution).
- What is SaMD utilizing AI for diagnosis of swallowing dysfunction?
-
If left untreated, age-related decline in oral function (called oral frailty) can lead to numerous physical and social disabilities, such as eating disorders and speech disorders, as well as generalized muscle weakness (frailty). Therefore, early diagnosis and appropriate treatment are important. Dysphagia, a type of oral function decline, is on the rise in an aging society, and it has been reported that approximately 70% of pneumonia deaths are caused by aspiration. Early detection of swallowing dysfunction and therapeutic interventions such as rehabilitation are important to prevent aspiration pneumonia. However, currently, the only swallowing assessment methods available are swallowing endoscopy and swallowing fluoroscopy, which place a significant burden on patients.
Since the organs used in swallowing and speech share many common parts, such as the tongue, oral cavity, and pharynx, we are focusing on the possibility of evaluating swallowing function from speech. We are developing AI that can evaluate swallowing dysfunction from speech data during conversation. In collaboration with several departments at Tohoku University (Otolaryngology, Dentistry, and Rehabilitation Department, School of Biomedical Engineering) and NEC Corporation, we have developed an AI system that analyzes the full frequency range of speech from patients visiting the Tohoku University Hospital Swallowing Treatment Center using an AI engine specialized in time-series data analysis (time-series model-free analysis). This system confirms the baseline of healthy subjects’ speech (gender, age, individual differences, etc.), detects differences between healthy subjects’ pronunciation and that of patients, and diagnoses swallowing dysfunction.
We will continue to train the system on data from elderly people with swallowing dysfunction and move toward practical application. If this program medical device is put into practical use, it is expected to enable easy and early diagnosis of patients with swallowing dysfunction who may develop aspiration pneumonia. We jointly filed an intellectual property application with Tohoku University in March 2023. Furthermore, we conducted a pre-development consultation with the PMDA in December 2023.
- What is SaMD utilizing AI for supporting respiratory function test?
-
Spirometry is a respiratory function physiological test that measures the amount of breath a subject exhales and the time it takes to exhale. It is an important test for diagnosing chronic obstructive pulmonary disease (COPD) and other lung diseases, but its widespread use is limited. This is because it requires the subject’s (patient’s) cooperation (effortful breathing), and it is difficult for non-specialists to determine whether the test was performed correctly and interpret the output results (flow-volume curve). Developing a system that allows even non-specialists to easily interpret results is considered an important medical challenge for diagnosing respiratory diseases and providing early treatment.
We are developing AI to interpret flow-volume curves in collaboration with Kyoto University and NEC Solution Innovators, Ltd. (NES). We have collected medical data from approximately 1,900 cases and are currently working toward practical application.
In July 2020, we entered into a joint development and commercialization agreement (license agreement) with Chest Co., Ltd., and received milestone payments in October 2021 and June 2023 based on the agreement with Chest, and received consideration for the exercise of option rights in February 2025.
- What other projects are you working on to develop SaMD utilizing AI?
-
As part of our efforts in women’s diseases, one of our priority areas, we are jointly developing AI with Tohoku University to detect lesions from breast cancer pathology images. In testing using pathology images, we classified the detection model into three classes (benign, non-invasive cancer, invasive cancer) or two classes (benign, malignant), achieving diagnostic accuracy of 88.3% and 90.5%, respectively. Going forward, we plan to also work on AI diagnosis using “intraoperative rapid pathology specimens” in the breast cancer field.
In addition, as part of our efforts in aging-related diseases, we are jointly developing AI with Tohoku University to predict the occurrence of arrhythmias and heart failure using information from cardiac implantable devices.
Furthermore, in September 2022, we signed a joint research agreement with Hi-Lex Corporation and Hi-Lex Medical Co., Ltd., and are working with Hi-Lex Medical Co., Ltd. and Tohoku University to develop AI to predict the occurrence of thrombosis in ventricular assist devices.
- What kind of collaborations do you conduct with NEC Corporation and NEC Solution Innovator, Ltd.?
-
Programmed medical devices that utilize AI require the development of core AI technology and the systems to run it in clinical settings. The optimal AI algorithm must be selected based on the medical issue to be solved and the type of medical data to be used.
We have licensed several basic AI algorithms (engines) from NEC Corporation (NEC) and customize them for medical use using medical data and advice from doctors in clinical settings. We have commissioned NEC Solution Innovators, Ltd. (NES) to develop the systems to utilize the medical AI we have developed in clinical settings.
- What is a disposable ultrafine endoscope?
-
Peritoneal dialysis patients constantly have a tube inserted into their peritoneum to inject dialysis fluid. In collaboration with Tohoku University, Juntendo University, and Jikei University School of Medicine, we developed an ultra-thin endoscope (approximately 1mm in diameter) that can be inserted through this thin tube to observe the peritoneal cavity non-invasively, without the need for open surgery or laparoscopy. In August 2022, an application for approval of the main fiberscope was submitted to the PMDA, and pharmaceutical approval was received from the Ministry of Health, Labour and Welfare in December of the same year.
In September 2022, we entered into a joint research agreement with Hi-Lex Corporation and its subsidiary, Hi-Lex Medical Co., Ltd., to develop medical devices, including the creation of the accessory guide catheter.
In May 2024, we entered into a licensing agreement with Hi-Lex Medical Co., Ltd. for this endoscope for medical uses, including peritoneal dialysis.
- What is the diagnostic for phenylketonuria?
-
Phenylalanine, one of the amino acids that make up proteins in the body, is metabolized by enzymes in the body to convert it to another amino acid called tyrosine. Phenyletonuria occurs when phenylalanine is not metabolized and accumulates in the body due to a congenital low enzyme activity. It is a pediatric disease designated as an intractable disease. Without appropriate treatment, this disease can lead to severe symptoms such as intellectual delay and seizures. Since the introduction of postnatal mass screening in 1977, almost all affected children have been diagnosed early.
However, affected children require a proper dietary regimen to restrict phenylalanine, and regular medical examinations are required. However, blood tests taken once every few months do not allow for detailed dietary management. Unlike diabetic patients, there are no home blood glucose monitoring systems, making self-management difficult.
We are collaborating with Tohoku University to develop a system that can easily and accurately measure blood phenylalanine levels at home. Our goal is to develop this new testing system into a kit and lead to insurance reimbursement for self-management. If patients with phenylketonuria could self-monitor their blood glucose levels at home at any time, like diabetic patients can do, it would enable them to manage their diet more precisely.
- What is important in the development of a medical device and software as a medical device?
-
Even with excellent technology, there are many cases where medical application (practical use) is difficult because the technology does not match the challenges and needs of the medical field or is inappropriate for the specifications of the medical field, and many companies with technology face this problem. Our company develops solutions to problems starting from the needs of the medical field, and develops optimized products by imagining the final product in the medical field, striving to develop medical devices and programmed medical devices based on the idea of “biodesign,” which realizes innovation.
Financials
- How were your full-year results for the fiscal year ending March 31, 2025?
-
For the full fiscal year ending March 2025, we achieved operating revenue of 132 million yen and net income of 113 million yen. In terms of revenue, in addition to contract fees with companies, we received a grant from the Japan Agency for Medical Research and Development (AMED), and additional milestone income was recorded, resulting in 147% of the plan.
In terms of expenses, the amount of operating loss was reduced compared to the initial plan by utilizing public funds from AMED in the development of pipelines such as medical devices for diabetes treatment support programs and chronic myeloid leukemia (CML).
Although we reduced our own research and development expenses, as a result of implementing efficient research and development activities, which are our strength, the initial target for pipeline development (number, stage) is progressing as planned.
In addition, as extraordinary income, we recorded cancellation fees associated with the termination of the license agreement with Baxter for disposable ultra-thin endoscopes and debt forgiveness gains associated with the reduction of debt for AMED’s CiCLE program, resulting in net income of 113 million yen.
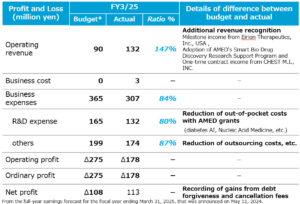
- When do you announce your financial results?
-
First quarter: around mid-August, second quarter: around mid-November, third quarter: around mid-February, financial results announcement: around mid-May.
Please see the following page.
- When is your business year and fiscal year end?
-
Our business year runs from April 1st to March 31st of the following year, and our accounting period ends in March each year.
- Where can I find the financial results?
-
Please see the following page.
- Where can I check past performance?
-
Please see the following page.
- Do you have a timeline for turning to profitability at this point?
-
It is difficult to say when we will be able to achieve stable profitability, but we achieved single-year profitability in the fiscal year ending March 2025. Going forward, we aim to achieve long-term profitability by actively utilizing public research grants and through income obtained from the out-licensing of our pipeline (lump-sum contract payments, milestone income paid according to the development stage, royalty income paid according to product sales, etc.).
Stock/Shareholder
- Who should I contact regarding shareholder procedures?
-
For inquiries regarding stock procedures, please contact the following:
Sumitomo Mitsui Trust Bank, Limited, Stock Transfer Agency Department
Tel: 0120-782-031
*If you have a securities account, please contact your securities firm.
- What is the status of your major shareholders?
-
Please see the following page.
- Who should I contact for IR inquiries?
-
We will post frequently asked questions in the FAQ section of our website.