Development status
Vitamin B6: RS8001
When we feel emotions or think about various things, “neurotransmitters” pass between neurons in the brain to transmit information. There are various types of neurotransmitters, including γ-aminobutyric acid (GABA), which has an anti-stress effect, and serotonin, which brings mental stability. When the neurotransmitters are out of balance, people can easily become stressed, irritable, and have other mental alterations. We have discovered the possibility that pyridoxamine (RS8001), a type of vitamin B6, can improve the balance of these substances and have been working to develop a therapeutic drug that can help women with mental health care.
Background of RS8001 development and the safety
The neurotransmitters GABA and serotonin have an amino group in their chemical structure. RS8001 (pyridoxamine), We develop a type of water-soluble vitamin B6 (pyridoxamine) that also has an amino group in its structure. Therefore, biochemical and animal studies suggest that pyridoxamine is utilized to produce GABA and serotonin and also inhibit the degradation of the neurotransmitters, resulting in the increment of these neurotransmitters in the brain. In fact, animals (rats) treated with pyridoxamine proved to be more stable in response to excitation and stimulation (stress). Pyridoxamine is a nutrient found in regular food and has been shown to be highly safe even when administered in large doses, but it is an unapproved medicine in developed countries, including Japan.
As society becomes more complex, many people live under stress, but compared to physical illnesses, medical treatment for mental illnesses is still insufficient. Medicines that control and regulate neurotransmitters may be useful in the treatment of such diseases. Today, women are expected to play an active role in society, and mental health care for women is an extremely important issue in society. We have been engaged in the research and development of the drug for the treatment of premenstrual syndrome/premenstrual dysphoric mood disorder, which makes social life difficult for women of reproductive age, and menopausal disorders during the menopause.
Therapeutic for menopausal disorder
Menopausal disorders are caused by the decrease in female hormone secretion and various psychological and social stresses during the perimenopausal period and involves various symptoms such as hot flashes (e.g., upper body flushing, glowing, sweating), mental symptoms (depression, anxiety, and insomnia), and physical symptoms (fatigue and joint pain), which interfere with daily life. Hormone replacement therapy, which supplements female hormone in small doses, is a typical drug therapy, but its use in Japan is limited to about 2% of the patients due to concerns about adverse events such as breast cancer.
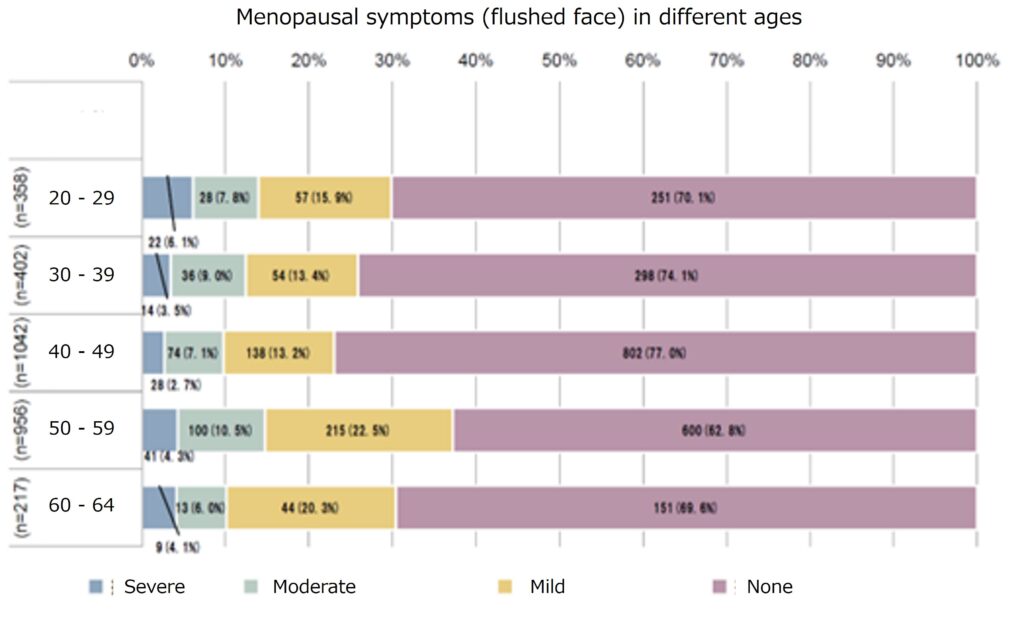
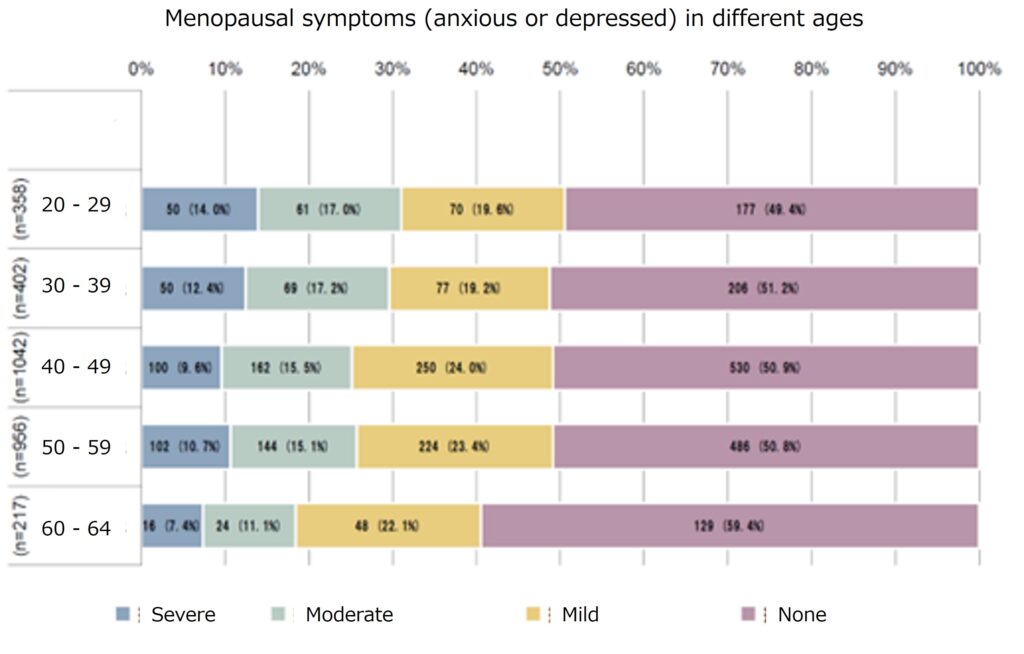
Our collaborators at Institute of Science Tokyo discovered that hot flashes and depression, the two major symptoms of menopausal women, were more severe in those with low vitamin B6 intake in the study on the relationship between various menopausal symptoms and nutrient intake, suggesting that vitamin B6 supplementation may alleviate these symptoms.
Therefore, we have been conducting a clinical study of RS8001 (pyridoxamine) as a treatment for hot flashes and depression in menopausal disorder. In December 2021, we signed a collaboration agreement with Institute of Science Tokyo. In March 2023, this project was selected for the AMED’s “Practical Research Project for Comprehensive Support of Women’s Health (the representative organization: Institute of Science Tokyo)”. This clinical research has been conducted in a double-blind design employing a placebo lead-in method with the aim of eliminating the placebo effect as much as possible.
Percentage of menopause disorder patients with hot flashes and depression