Development status
Software as a Medical Device (SaMD) Utilizing Artificial Intelligence for Supporting Respiratory Function Test
The World Health Organization (WHO) considers respiratory diseases as important noncommunicable diseases (NCDs) in addition to cancer, diabetes, and cardiovascular diseases. Typical respiratory diseases include chronic obstructive pulmonary disease (COPD) and asthma. COPD is also highlighted as an important disease in the revision of “Health Japan 21” by the Ministry of Health, Labour and Welfare, and is referred to as a “lifestyle disease of the lung”.
A typical respiratory function test for the diagnosis of COPD and other lung diseases is a pulmonary function test (spirometry), which measures the amount of breath a subject exhale and the duration of exhalation.
Current Diagnosis and Challenges
In addition to requiring the effortful cooperation of subjects (patients) (effort breathing), spirometry diagnosis requires determining whether the test is performed correctly and interpreting the output results (flow volume curve). However, because interpretation is difficult for unexperienced physicians, spirometry is not widely used.
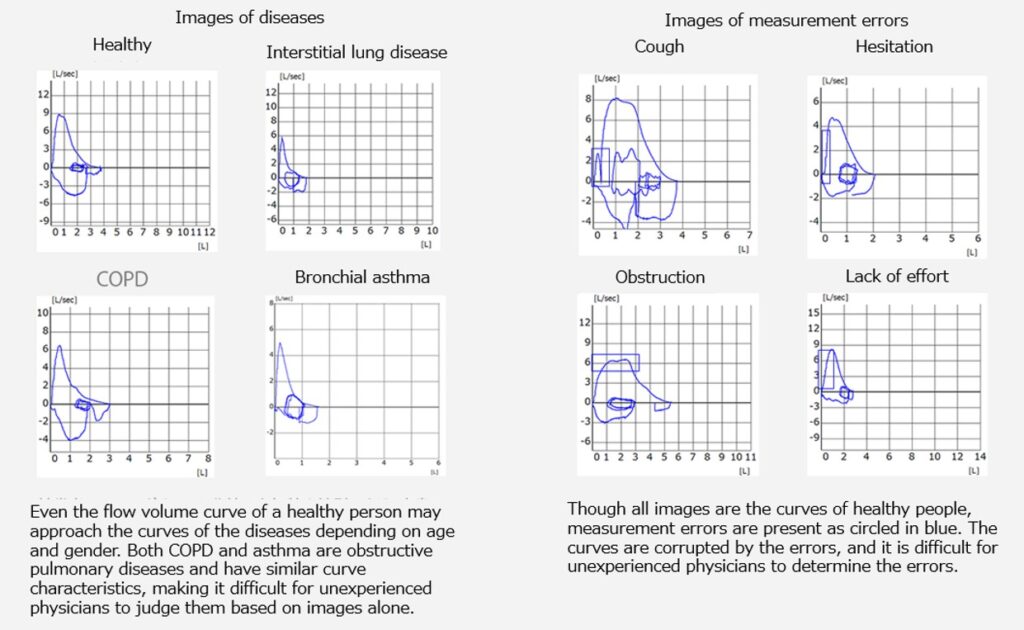
To solve issues in the medical setting in the respiratory field, we have decided to develop software as a medical device (SaMD) by utilizing artificial intelligence (AI) that detects diseases and measurement errors from images that are difficult to interpret.
Results to Date
Initial Verification
The development of the SaMD requires that issues in the medical setting be obtained from medical professionals and that the most appropriate AI program be selected based on the utilization method and on the data that can be obtained. In the initial verification, we considered two methods: “image analysis” using flow volume curve images discharged from diagnostic equipment of respiratory function test, and “numerical data” stored in the equipment. Therefore, we analyzed which method could be expected to provide higher classification accuracy by utilizing a small amount of data.
As a result of the analysis, even experienced pulmonologists found it difficult to properly label disease characteristics in “numerical data” and we could not achieve the expected classification accuracy in the “numerical data” method, while a certain level of classification accuracy was achieved and appropriate labeling was possible in “image analysis”. Based on the results of this validation, we decided to utilize AI to classify diseases and errors from images.
Development Stage
The targets for the classification were determined as follows, after discussion with the pulmonologists and Chest MI Inc.
Disease Category “Normal/Bronchial Asthma/COPD/Interstitial Lung Disease”
Measurement Error “Cough/Hesitation/Obstruction/Lack of Effort
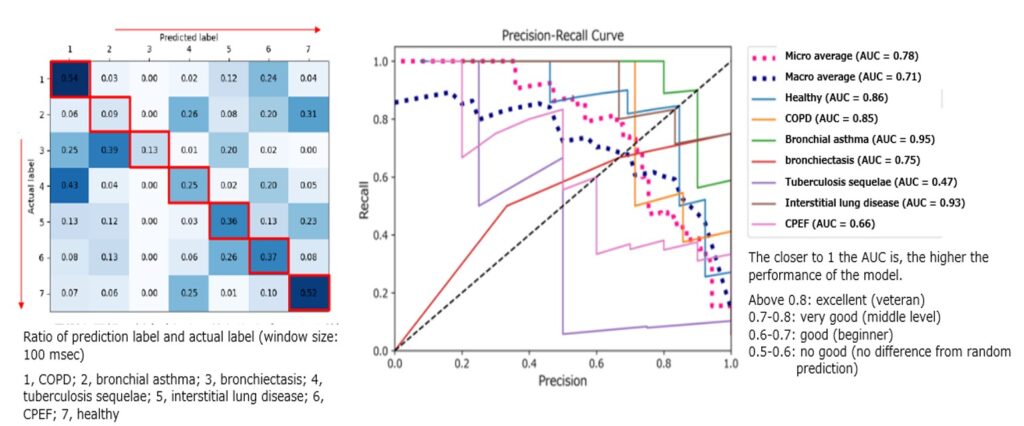
The validation of the initial accuracy for determining the optimal predictive AI algorithm was performed using spirometry image data from 1,900 cases. Then, after building a dataset of correct cases, seven disease data sets were analyzed: COPD, bronchial asthma, idiopathic pulmonary fibrosis (IPF), healthy subjects, bronchiectasis, emphysema complicated pulmonary fibrosis (CPFE), and tuberculosis sequelae. As a result, the results using spirometry image data were 86% (healthy subjects), 95% (bronchial asthma), 85% (COPD), 93% (IPF), 75% (bronchiectasis), 66% (CPFE), and 47% (tuberculosis sequelae), respectively, and the accuracy was high enough for each case. We competed the development stage in March 2023, and agreed to move to the commercialization stage with Chest MI Inc. in June 2023. Chest Corporation will take the lead in the development of systems and other aspects of practical application.