News
Announcement of Publication of an Overview of Artificial Intelligence (AI) Supporting Safe and Secure Maintenance Hemodialysis
In collaboration with Tohoku University, NEC, and others, our company is developing a software as a medical device (SaMD) equipped with artificial intelligence (AI) to support hemodialysis treatment. Clinical performance test1) to evaluate the device’s clinical usefulness was conducted at eight domestic facilities, including St. Luke’s International Hospital, and the results demonstrated that the device has predictive accuracy equivalent to that of dialysis specialists (non-inferiority) (disclosed on October 20, 2025). We are pleased to announce that an overview of this SaMD was published in the February 2026 issue of “Clinical Dialysis.”
Chronic hemodialysis patients have impaired renal function, making it difficult to remove fluid and waste products from the body. Managing fluid removal (water removal) is one of the most important goal of hemodialysis treatment. Insufficient water removal poses a risk of damaging the patient’s cardiopulmonary function. On the other hand, excessive water removal can cause hypotension during dialysis, potentially leading to serious adverse events such as nausea and loss of consciousness. Therefore, determining the appropriate amount of water removal requires expertise and is a major challenge for physicians. Currently, dialysis facilities lack a sufficient number of specialists, and in rural areas and at night, non-specialists, experienced nurses, and clinical engineers assist in setting the amount of fluid removal. To address this lack of human resources and ensure safe and reliable dialysis treatment anywhere, there was an urgent need to develop a programmable medical device that utilizes AI to mimic the “experiential knowledge (tacit knowledge)” of experienced dialysis specialists.
This SaMD is equipped with a proprietary AI engine, “DCCN (Dual-Channel Combiner Network),” jointly developed by Tohoku University and NEC Research Laboratories America. DCCN is based on deep learning and is a groundbreaking AI that can directly integrate time-series data with different sampling rates (acquisition intervals), such as “dialysis information three times a week,” “blood test results twice a month,” and “patient profiles,” for learning and prediction. The AI is trained on a massive amount of dialysis data, totaling approximately 400,000 sessions performed by dialysis specialists. Based on information from the past five dialysis sessions, as well as information before the day’s dialysis, the most recent blood test results, and the patient profile, it determines the appropriate amount of fluid removal.
With support from the Japan Agency for Medical Research and Development (AMED), we conducted a multi-center clinical performance test of this SaMD at eight medical institutions nationwide, including St. Luke’s International Hospital and Kawashima Hospital. This test compared the target fluid removal volume prescribed by dialysis specialists with the AI-predicted fluid removal volume for 108 clinical cases performed by dialysis specialists. The primary endpoint, agreed upon in consultation with the Pharmaceuticals and Medical Devices Agency (PMDA), was set at an accuracy rate of 80%, which is the acceptable range of error between the AI prediction and the specialist’s setting. The AI achieved an average accuracy rate of 90.0%, far exceeding the target and achieving excellent results. For a mean fluid removal volume of 2,353 mL, the mean absolute error (MAE)2) between the AI and the specialist’s prediction was only 119.2 mL (an error of about one cup), and the mean absolute Percentage Error (MAPE) 3)was only 5.2%. These results demonstrate that this AI-SaMD is non-inferior (equivalent) to dialysis specialists in setting water removal volumes. Furthermore, consistently high accuracy was demonstrated across multiple geographically diverse facilities, confirming its high versatility.
The practical application of this SaMD is expected to significantly reduce the burden on medical professionals involved in dialysis treatment with limited human resources, enabling the provision of safe, high-quality dialysis treatment at a specialist level anywhere in the country. Furthermore, we are developing a next-generation technology called “P-DCCN (Personalized DCCN),” which continuously learns from individual patient data to enable predictions that are more optimized for each patient’s characteristics. In the future, we hope to contribute to the realization of “personalized medicine,” which proposes optimal water removal plans for each patient throughout their lifelong dialysis treatment.
An overview of this programmable medical device was published in the February 2026 issue of “Clinical Dialysis.”
1) Clinical performance test
In order to enable the use of a software as a medical device (SaMD) under development in the medical field, it is necessary to confirm whether the software as a medical device will perform as expected in the clinical field using actual human clinical data. Clinical performance tests are clinical studies conducted for this verification. Based on the performance confirmed in the clinical performance test, an application (pharmaceutical application) will be submitted to the Ministry of Health, Labor and Welfare to manufacture and sell the device as a software as a medical device. This clinical study has a similar nature to a validation test (Phase III trial) for pharmaceuticals.
2) MAE (Mean Absolute Error)
MAE is one of the indicators for evaluating the accuracy of a predictive model. Specifically, it indicates the average absolute value of the difference between the predicted value and the actual value. MAE is calculated as follows.
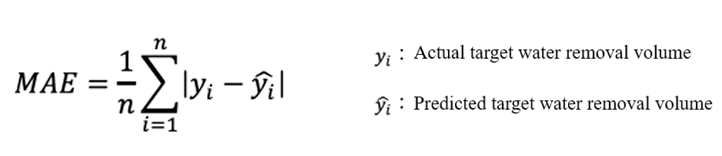
The smaller the value, the more accurate the prediction.
3) MAPE (Mean Absolute Percentage Error)
MAPE is one of the indices for evaluating the accuracy of a forecast model. Specifically, it is the average percentage of error relative to the actual value. MAPE is calculated as follows.
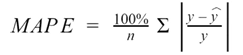
The smaller the value, the more accurate the prediction.