Business Model
Our research and development strategies
1) Development of various modalities (pharmaceuticals, medical devices, and programmed medical devices using artificial intelligence (AI))
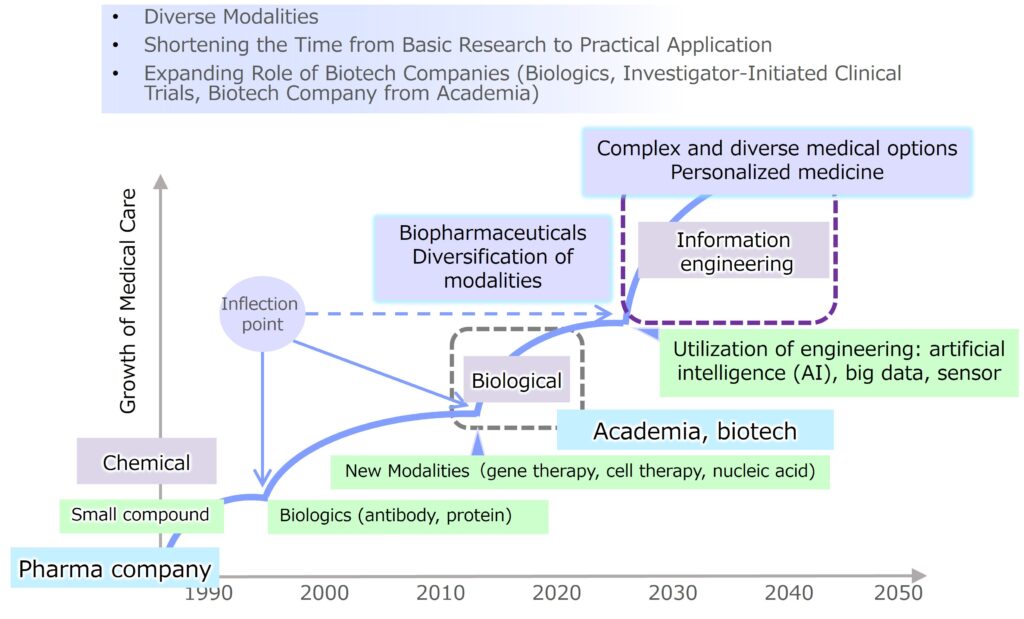
Our company wishes to solve medical issues and contributes to medical innovations that allow people to enjoy health throughout their lives, both physically and mentally. Drug development is expanding from small molecule drugs to various modalities such as biopharmaceuticals (antibody drugs, nucleic acid drugs, gene therapy, cell therapy). Furthermore, with recent advances in engineering and information technology, we are seeing a shift to new medical care through the fusion of engineering and information technology. Medical treatment options are expanding, including pharmaceuticals, medical devices, and even programmed medical devices using artificial intelligence (AI). Therefore, in addition to chemical and biological researches, we are broadening our horizons to include engineering and information technology researches, creating a diverse and attractive research and business portfolio.
2) Solving medical issues for declining birthrate and aging society/longevity
In developed countries including Japan, the super-aging society is progressing, and the difference between average life expectancy and healthy life expectancy (the period during which one can live independently and be healthy both physically and mentally, minus the period of nursing care such as being bedridden or having dementia) is about 10 years, which is a major medical and social issue. If we can overcome various aging-related diseases, such as cancer, cardiovascular disease, respiratory disease, and diabetes, it can lead to an extension of healthy life expectancy. These four diseases account for 70-80% of the world’s deaths, and the World Health Organization (WHO) positions them as important diseases associated with aging and lifestyle habits, ‘non-communicable diseases (NCDs).’ Our mission is working to extend healthy lifespan and to provide new therapies for women’s and children’s diseases.
-1.png)
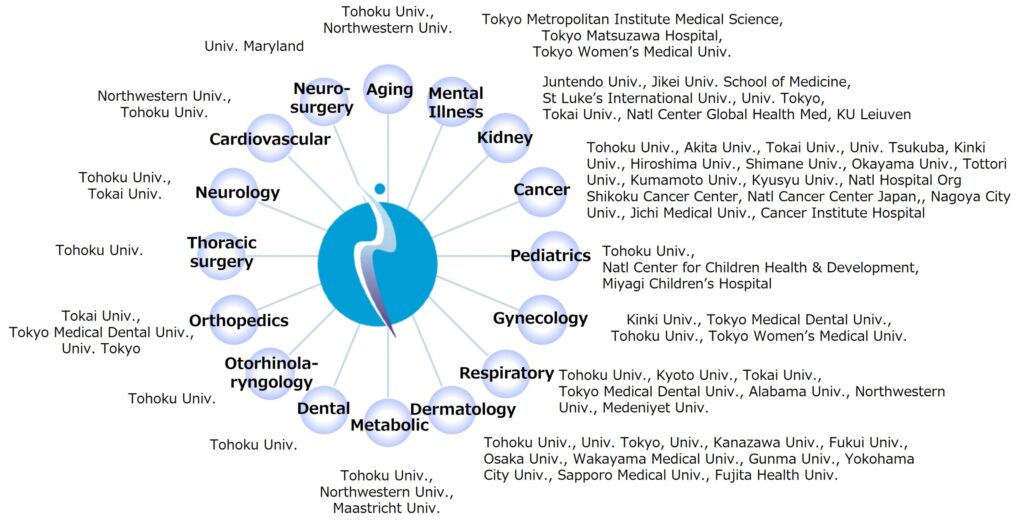
3) Network with research institutes and medical institutions
The roles of public research institutes and medical institutions such as university hospitals in creating medical innovation are also expanding. Unlike traditional small molecule drugs, the basic technologies and seeds of biopharmaceuticals using genetic engineering, etc. are oowned by public research institutions. In addition, medical institutions own the medical big data necessary for developing program medical devices using artificial intelligence (AI). Since our company conducts multiple investigator-initiated clinical trials with the medical departments of many medical institutions, we are in an environment where it is easy to access the medical data necessary for understanding medical issues and developing AI. Rather than sticking to our own resources and research environment, we would like to focus on actively utilizing external resources and external environments (networks with public research institutes and medical institutions) and build a framework for efficiently creating innovation.
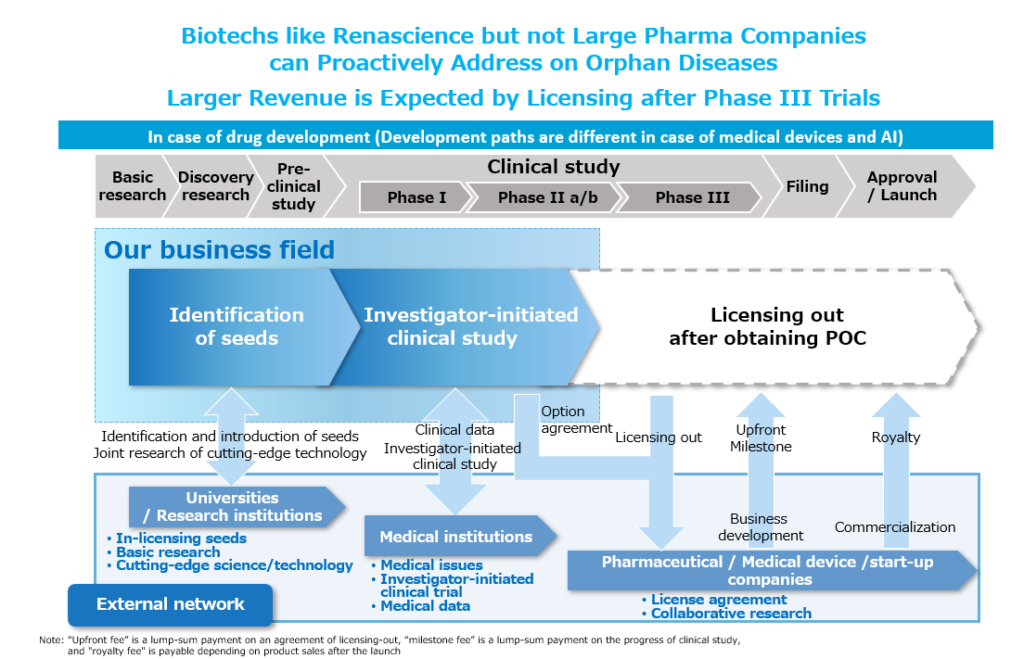
4) From basic research to investigator-initiated clinical trials
Our company conceives treatment concepts and ideas from basic research and manufactures new pharmaceuticals, medical devices, etc. We would like to complete appropriate non-clinical studies (animal testing), ultimately verify our concepts in human clinical trials, and develop them in-house to the stage where they are close to applying for regulatory approval. For example, the ultra-fine endoscope, a medical device approved in December 2022, was developed in collaboration with several universities from product development to the implementation of non-clinical studies and the completion of investigator-initiated clinical trials, and was applied for regulatory approval after being licensed to a medical device company. In addition, the new pharmaceutical for chronic myeloid leukemia, a blood cancer, is currently undergoing phase III trials, which are the final clinical trial required for regulatory approval application, but we would like to complete this trial in-house. We also conduct some clinical performance tests for regulatory approval for program medical devices that utilize artificial intelligence (AI). By developing in-house to the stage close to regulatory approval, it is possible to reliably implement important seeds to commercialization. In addition, licensing out a seed at a later stage of development will enable us to receive a larger compensation, which will enable us to return a larger share to our shareholders.
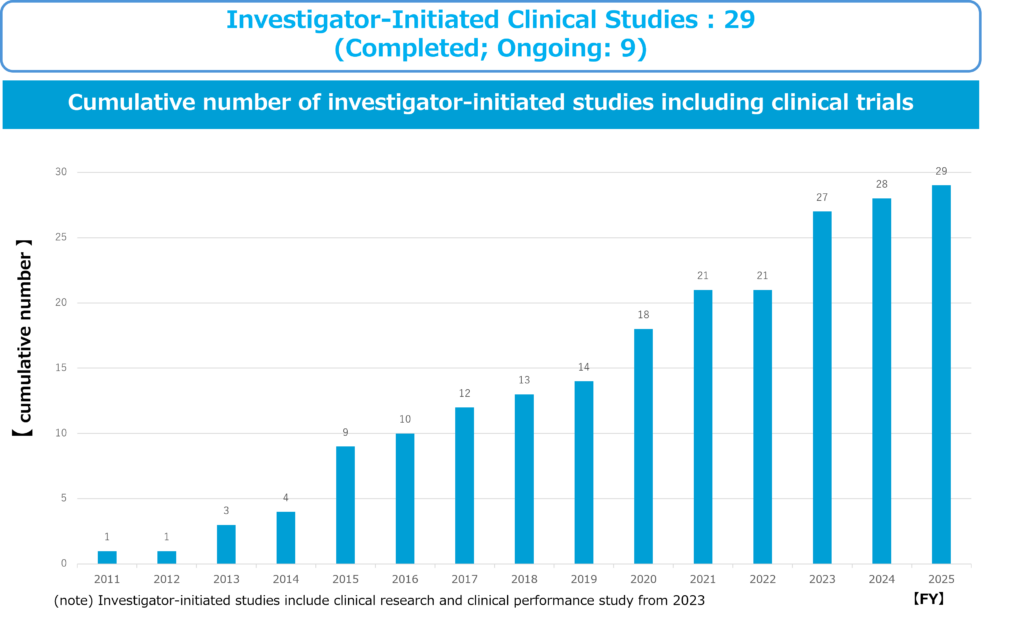
5) Investigator-initiated clinical trial
Our company has thus conducted 29 investigator-initiated clinical trials led by physician scientists. The advantages of investigator-initiated clinical trials are quality and speed, i.e. efficiency. In investigator-initiated clinical trials, doctors who are at the forefront of research and can come into contact with the latest research results and who treat patients daily in the medical field can register appropriate patient subjects and decide trial plans. Since doctors themselves can conduct clinical trials, this is a suitable clinical trial framework for exploratory clinical trials of unapproved drugs. In addition, development of treatments for orphan diseases (since there are few patients, sales cannot be expected) is not initiated by pharmaceutical companies due to low profitability, so there are cases where investigator-initiated clinical trials must be conducted from start to finish. Almost the majority of drug development costs are spent at the clinical development stage, not the basic research stage. Investigator-initiated clinical trials can reduce development costs by quickly utilizing the scientific and technological achievements of cutting-edge universities and allowing clinical trial physicians to appropriately select patients who will be treated. If an appropriate clinical trial coordinating physician is found and support from multiple large medical institutions such as universities is obtained, investigator-initiated clinical trials have a great advantage over corporate clinical trials, and clinical trials can be conducted in a short period of time.
The 2003 amendment to the Pharmaceutical Affairs Law opened the way for investigator-initiated clinical trials in which doctors themselves conduct clinical trials, but it is still difficult for doctors to prepare new invetigational drugs necessary for clinical trials, including safety tests and formulations. At the time of the amendment, there were many new drugs approved overseas but not approved in Japan, and drugs used off-label (so-called drug lag), so drugs not approved in Japan and drugs used off-label were the mainstream tested in investigator-initiated clinical trials. In terms of ease of conducting clinical trials (existing data can be used from manufacturing to safety tests), doctors at many medical institutions such as universities have been involved in investigator-initiated clinical trials of new drugs approved overseas (not approved in Japan) and drugs used off-label. In addition, there were also cases where investigator-initiated clinical trials were conducted using existing drugs for orphan diseases that pharmaceutical companies do not address. Due to these backgrounds, there may be still a strong impression that “investigator-initiated clinical trials are for drugs approved overseas (not approved in Japan), the expansion of indications of existing drugs, and orphan diseases.” However, all clinical trials conducted by our company are for unapproved drugs (first-in-human), and are not clinical trials for expanding indications of overseas approved (not approved in Japan) drugs or existing drugs. In our drug development, non-clinical trials are conducted in compliance with GLP (Good Laboratory Practice, standards for the implementation of drug safety), and investigational drug manufacturing is conducted in compliance with GMP (Good Manufacturing Practice, standards for the manufacturing management and quality control of investigational drugs). In addition, investigator-initiated clinical trials are conducted in compliance with GCP (Good Clinical Practice, standards for the implementation of clinical trials of drugs), just like corporate clinical trials. Therefore, they can be used for regulatory approval applications. In addition, our drugs are unapproved drugs and we have secured intellectual property, so we can commercialize them exclusively.
6) Ecosystem based on open innovation
Many pharmaceutical companies and drug discovery bio start-ups have focused on building their own pipeline value chains (accumulating all the development processes) and increasing business value. However, in fields such as pharmaceuticals, where the success rate is extremely low, the development period is long, and investment is large, the research and development and business risks are high, so it is essential to form a portfolio that combines many pipelines and diversify the risks. Our company has been practicing development to improve development efficiency, including costs, by utilizing the resources of external institutions (research institutions, medical institutions). We are considering building many value chains based on alliances with external institutions, and our strategy, research and development, and human resource management are different from existing bio start-ups. We have expanded many pipelines with few human resources and expenses, and have been able to develop modalities, so we are beginning to see outcomes. Rather than focusing only on our own resources and the internal environment, we would like to focus on actively utilizing external resources and the external environment and build a framework that efficiently creates innovation.
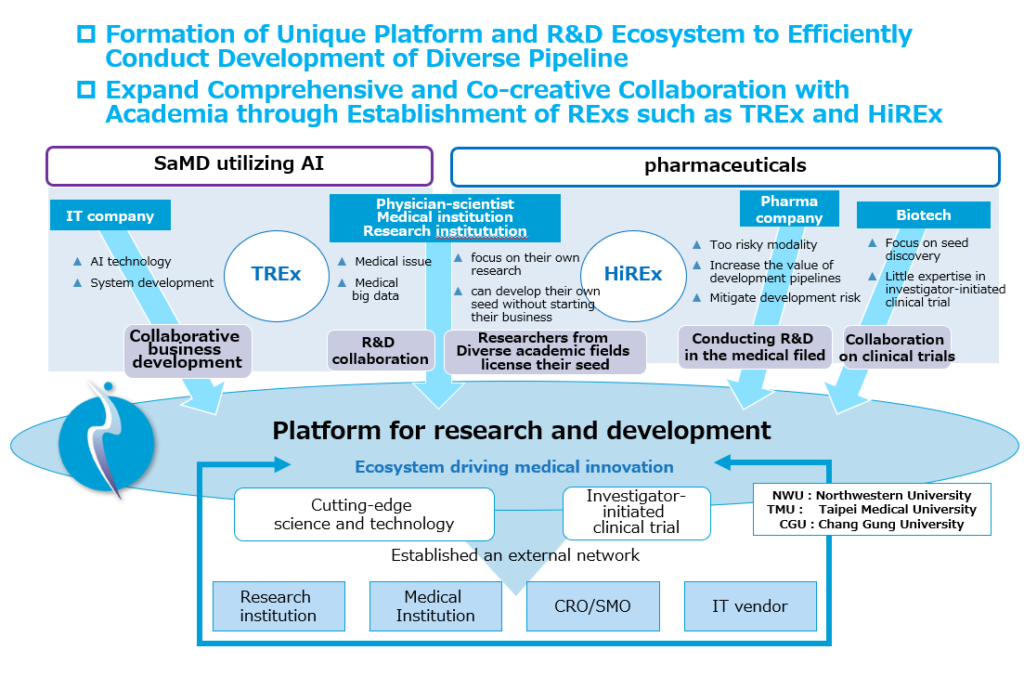
7) Collaborative researches with multiple universities through the opening of the Renascience Open Innovation Lab (REx)
Our company believes that there is a need for a place to utilize cutting-edge scientific and technological achievements, a place for face-to-face interaction with doctors and researchers, and a place for open innovation also with government and medical industry companies. In January 2022, we opened the Tohoku University Renascience Open Innovation Lab (TREx) at the Medicinal Hub of the Graduate School of Medicine of Tohoku University (2-1 Seiryo-cho, Aoba-ku, Sendai, Miyagi Prefecture, Faculty of Medicine Building No. 5). TREx is also the first project to be located at a base based on the “Agreement on Regional Economic Development between Sendai City and Tohoku University” concluded in April 2021. In addition, in April 2023, our company signed a comprehensive collaboration agreement with Hiroshima University and opened the Hiroshima University Renascience Open Innovation Lab (HiREx). In October 2024, we signed a joint research agreement for pharmaceuticals and program medical devices with Taipei Medical University (TMU)-Biotech, and in January 2025, we agreed to establish a laboratory for longevity research within TREx with Northwestern University’s Potocsnak Longevity Institute. We will continue to conduct joint research based on open innovations with many public research institutes involved in cutting-edge research both in Japan and overseas.

